發表文章
Journal Papers
期刊
2025
-
Reversibly interconverted Cu+/Cu+-H species as active sites for selective hydrogenation of fatty acid methyl esters to fatty alcohol over layered double hydroxide derived CuMgAlOx catalysts
ChanisaraNootoa,PanaleeChuaykaew,PawanratSingthuena,ThanasakSolos, YardthipPreedawichitkun, KanyanatKhosukwiwat, KanokwanWengwirat, PratyaPromchana, RajuKumar, Po-Wen Chung, YingyotPoo-arporn, WanwisaLimphirat, KittisakChoojun,* ,TawanSooknoi,*Molecular Catalysis 2025 575, 114898High fatty alcohol production (>90 % selectivity) can be achieved though the selective hydrogenation of fatty acid methyl esters over layered double hydroxide derived CuMgAlOx catalysts in a fixed-bed reactor at 250 °C under atmospheric H2. ∼17 wt.% Cu loading CuLDHs with different Mg2+/Al3+ ratios (CuMg60Al40Ox, CuMg70Al30Ox, CuMg75Al25Ox, and CuMg80Al20Ox) were prepared by co-precipitation-hydrothermal method. The Cu dispersion and species were determined by H2-TPR, consecutive H2-TPR, N2O-dissociative reaction, and in situ TR-XANES. Highly dispersed Cu metal along with cationic Cu(I) species were obtained for all CuMgAlOx. The cationic Cu(I) species (Cu+/Cu+-H) content increased with Mg2+ content. In the presence of H2, the cationic Cu(I) species undergo reversible interconversion between Cu+ and Cu+-H species, facilitating hydrogen dissociation/evolution. The hydrogenation activity was governed by the balance of the metallic Cu surface and the cationic Cu(I) species. The Cu+ species allow preferential adsorption of Cdouble bondO ester for selective hydrogenation of FAMEs to fatty alcohol (>90 % selectivity). With Cusurface/Cu(I) ratio at 0.25 (CuMg75Al25Ox), the fatty alcohol production rate of 49.3 h−1 was obtained with high stability due to the reversible interconversion of Cu+/Cu+-H that prevented product re-adsorption and side reactions.
-
Stabilized Pd Nanoparticles Encapsulated in MIL-101(Cr) forChemoselective Hydrogenation of Polyunsaturated FAMEs.
Yardthip Preedawichitkun, Natthida Numwong, Narong Chanlek, Po-Wen Chung, Raju Kumar, Chularat Wattanakit, Warot Prasanseang, Pratya Promchana, Chanisara Nooto, Phuwadon Khenkhom, Kittisak Choojun,* and Tawan Sooknoi*ChemCatChem 2025 17 (22), e00985Palladium nanoparticles (Pd NPs) were successfully encapsulated within the porous framework of MIL-101(Cr) via a double solvent method to produce highly dispersed and stable catalysts for the chemoselective hydrogenation of polyunsaturated fatty acid methyl esters (FAMEs). Pd loadings ranging from 0.5 to 1.5 wt.% were systematically studied to elucidate the effects of nanoparticle size, dispersion, and hydrogen activation behavior on catalytic performance. The 0.8Pd/MIL-101(Cr) catalyst exhibited the highest turnover frequency (TOF ∼9,700 h−1) and superior selectivity (>90%) toward monounsaturated products (C18:1), attributed to optimal Pd dispersion. In contrast, the 0.5Pd/MIL-101(Cr) showed an induction period under low H2 partial pressure, indicating limitations in hydride accommodation, while the 1.5Pd/MIL-101(Cr) suffered from Pd aggregation, resulting in a reduced intrinsic activity. Product selectivity was primarily governed by overall conversion: C18:1 was favored at low conversions, whereas C18:0 formation increased at higher conversions due to secondary hydrogenation. The catalysts demonstrated excellent stability and recyclability over multiple cycles without detectable Pd leaching or structural degradation. These findings establish MIL-101(Cr) as a robust and tunable platform for dispersing Pd NPs and highlight the potential of Pd/MIL-101(Cr) catalysts for efficient, selective upgrading of bioderived feedstocks under mild reaction conditions.
-
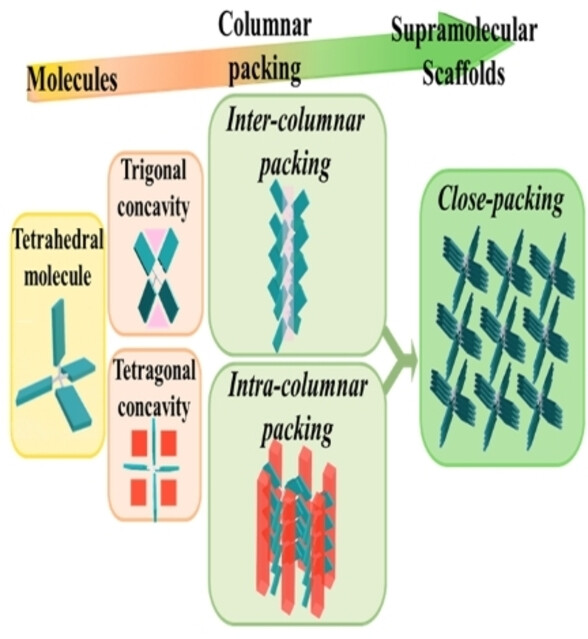
The Roles of Molecular Concavities in the Hierarchical Self‐Assembly of Giant Tetrahedra for CO2 Uptake
Heng‐Yi Lin , Huan‐Chang Lu, Chih‐Hsuan Tsai, Hsueh‐Ju Liu, Po‐Weng Chung, Wei‐Tsung Chuang,* Wang Chien‐Lung*Chemistry - A European Journal 2025 31, e202403348Giant tetrahedral molecules have sparked significant interest in the past decade due to their unique and diverse supramolecular nanostructures. The longer and bulkier peripheral substituents create deep molecular concavities and thus contribute to the different self-assembly behaviors compared to the conventional small tetrahedral molecules. In this study, a molecular giant tetrahedra, TetraNDI, was synthesized to investigate the important roles of the molecular concavities in the self-assembly mechanism. Single-crystal structural characterizations indicate that the TetraNDI takes its trigonal concavities to form 1D supramolecular columns, and its tetragonal concavities to reach close inter-columnar packing. The difficulty in occupying the concavities leads to the path-dependent phase behaviors of the giant tetrahedra. It is also found that the remaining molecular concavities in the supramolecular scaffolds affect the CO2 affinity of TetraNDI. With an understanding of the packing principles of molecular giant tetrahedra, the structure-property relationships could be better evaluated in the future and might broaden the horizon of porous materials.
-
具原子分散性之水滑石氧化物負載銅觸媒的製備與鑑定及其於生質能轉換的探討
Shiou-Min Wu (巫秀敏), Chih-Hsuan Tsai(蔡芷瑄), Raju Kumar(庫拉育), Po-Wen Chung(鍾博文)*材料世界網 2025/03/17
2024
-
觸媒化學於生質化學品生產之應用
(Application of Catalysis in Bio-based Chemical Production)陳冠豪(Kok-Hou Tan), 巫秀敏(Shiou-Min Wu), 鍾博文(Po-Wen Chung)*化學 2024 82, 195自工業革命以來,溫室氣體(GHG)排放量不斷的激增導致全球暖化,已經威脅到地球生命的生存環境。為了限制氣溫上升和造成破壞性氣候變遷的影響,世界已同意並計劃在2050年實現淨零排放。在杜拜舉行的COP28會議後,與會者承諾抽離對石化資源的依賴,使2035年時溫室氣體排放量要比2019年減少60%。除了要廣泛地促進碳捕集、利用與封存(CCUS)技術外,還需要致力於利用節能、可再生資源生產替代傳統工業流程中的石化原料,並透過降低燃料的消耗來減少碳足跡,確保供應鏈免受石油耗竭的威脅。因此,使用有效的觸媒進行高效催化反應,是促使產業成功轉型以實現永續發展目標的關鍵。我們總結了我們實驗室在催化木質纖維素和甲殼素衍生物轉化為C4和C6增值生質化學品方面的工作,以及概述了我們在新型有效觸媒及其反應機制方面的發現。研究證明了這些高效率的催化製程在生質工業化學品永續生產中具有很大的潛力,這對於實現淨零目標至關重要。
並列摘要
The surge in greenhouse gas (GHG) emissions since the Industrial Revolution has caused global warming which is threatening the existence of life on Earth. To limit the rise in temperature and the effect of destructive climate change, the world has agreed and set to achieve net-zero emissions by 2050. After the COP28 in Dubai, the participants have committed to shifting from relying on fossil resources and to achieving 60% of the GHG emissions cut, with respect to the level in 2019, by 2035. In addition to widely promoted carbon capture, utilization, and storage (CCUS) technology, efforts have been devoted to replacing fossil-based raw materials in conventional industrial processes with energy-efficient, renewable sources-based production to reduce carbon footprint through lowering fuel consumption and secure the supply chain from depleting fossil resources. An efficient catalytic reaction with an effective catalyst is, therefore, the key to the successful transformation of the industry to meet its sustainability goal. Here we summarize our works on the catalytic conversion of lignocellulose and chitin derivatives into value-added C4 and C6 bio-based chemicals, which outlines our findings on the novel effective catalysts and their reaction mechanism. These studies demonstrated the great potential of efficient catalytic processes in the sustainable production of bio-based industrial chemicals that are vital to the race to the net-zero goal.
-
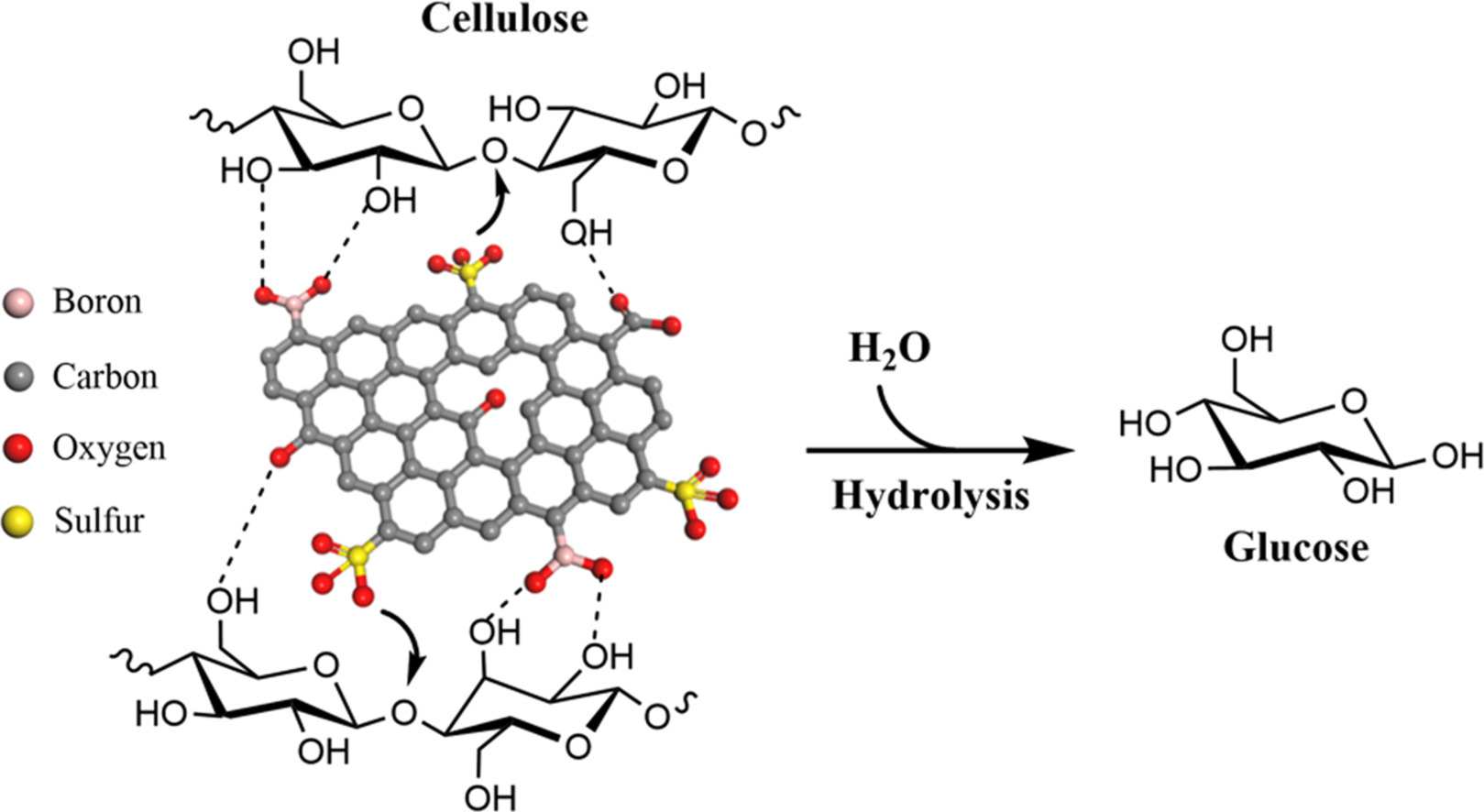
Hydrolysis of Cellulose using boronic-acid multifunctional carbocatalysts
Gemechu Dadi Bedasso, Chan-Yi Lin, Chia-Hui Wu, Der-Lii M. Tzou, Po-Wen Chung*Journal of the Taiwan Institute of Chemical Engineers 2024, 157, 105435Background Developing
Effective and sustainable catalysts for cellulose hydrolysis is crucial for transforming this abundant biopolymer into renewable chemicals and biofuel. Carbonaceous solid acids are promising catalysts for this reaction due to their low cost, chemical and hydrothermal stability, tunable surface, and high affinity for cellulose.
Methods
We developed multifunctional carbocatalysts for cellulose hydrolysis by introducing boronic groups and weak acid sites along with high-density sulfonic acid through borylation of mesophase pitch carbon (MPC) followed by sulfonation. The functionalized MPC was characterized using various techniques, including FT-IR, XPS, SS-NMR, Raman spectroscopy, and TGA.
Significant findings
The results of surface characterization revealed the successful introduction of multifunctional groups on the peripheral surface of MPC. This carbocatalyst can effectively hydrolyze pretreated cellulose into glucose, resulting in a maximum yield of 64 % in water at 180 °C for 3 h. Boronic groups and weak acid sites could mediate the interaction of cellulose molecules with catalyst surface to enhance the catalytic hydrolysis. The apparent activation energy for breaking the glycosidic bonds using the functionalized MPC is 99 kJ/mol, which is lower than the one derived from homogeneous acids. Furthermore, the carbocatalyst is recyclable at least four cycles prior to the significant loss for reactivity.
2023
-
淺談生質化學品的未來與意義The Roadmap and Significance of Biobased Chemicals
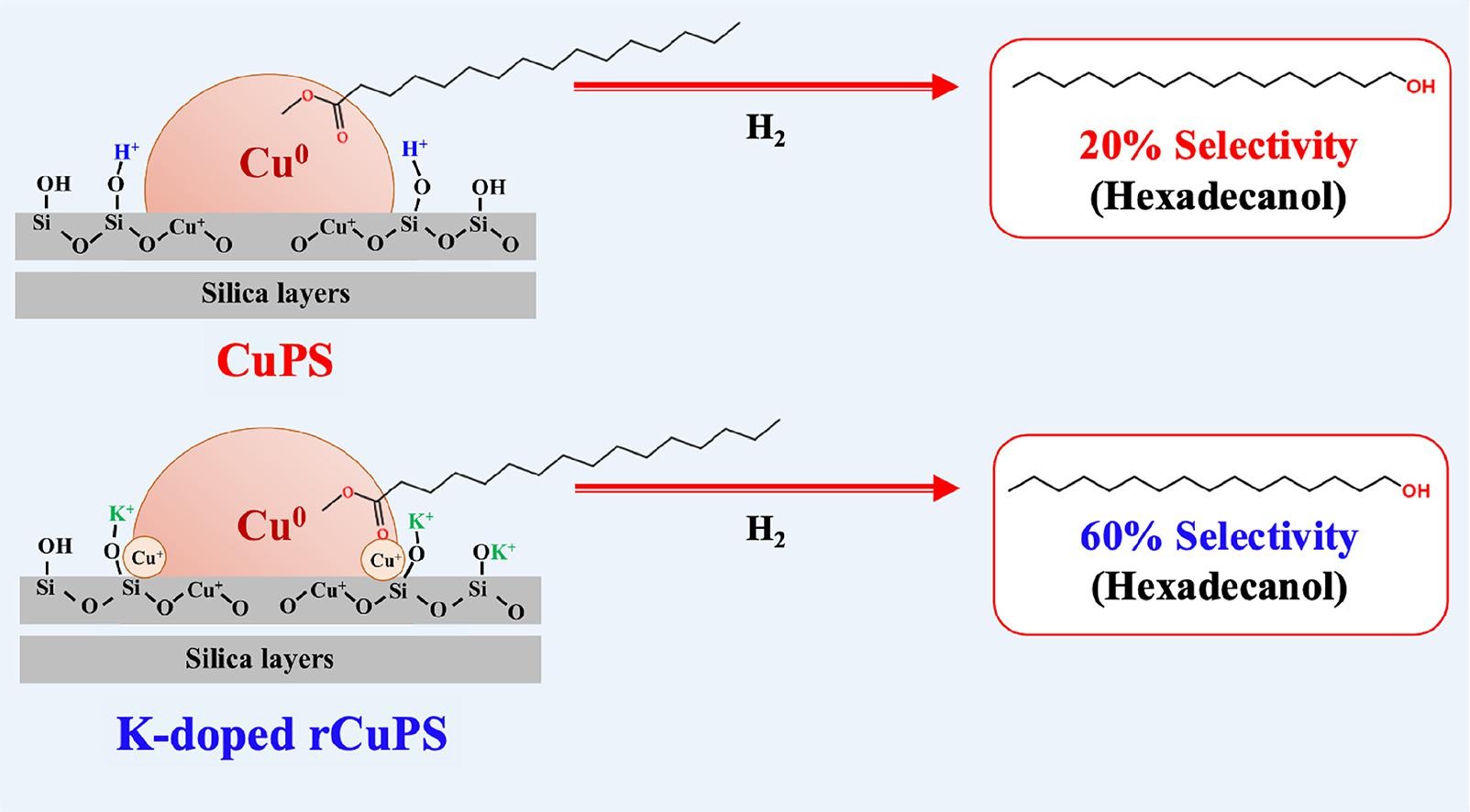
Po-Wen Chung(鍾博文)IEK產業情報網 2023/10/19Tuning Cu+ species/Brønsted acids of copper phyllosilicate by K+ doping for selective hydrogenation of methyl palmitate to hexadecanol
Warot Prasanseang, Kittisak Choojun*, Yingyot Poo-arporn, Ai-Lin Huang, Yu-Chuan Lin, En Chen, Hsin-Hui Lee, Po-Wen Chung, Tawan Sooknoi*Journal of Catalysis 2023, 428, 115115Selective hydrogenation of methyl palmitate to hexadecanol can be manipulated by tuning Cu+ species and Brønsted acid sites (BAS) of copper phyllosilicate (CuPS) catalysts with K+ doping. The catalysts were prepared by impregnating K+ onto reduced and non-reduced CuPS. The reactions were carried out in a fixed-bed flow reactor at 250 °C under atmospheric H2. In situ TR-XANES and Py-IR suggest that the presence of K+ could stabilize Cu+ species and neutralize BAS. As compared to the non-reduced sample, K+ loading (0.01–0.10 wt%) on the reduced CuPS provide higher Cu+ fraction (10–16%), lower BAS (0.82 to 0.16 mol/g) and lower Cu dispersion (75 to 52%). A balance between Cu0 active surface and Cu+ content provides an optimum hydrogenation activity (up to 80 %). The increased Cu+ species, together with the decreased BAS, does not only enhance the catalyst stability, but also hexadecanol selectivity (from 35 to 60%, at ∼50% conversion).
-
Amino group functionalized pitch-based carbocatalyst for the Henry reaction of furfural
Gemechu Dadi Bedasso, Der-Lii M. Tzou, Po-Wen Chung*Journal of the Taiwan Institute of Chemical Engineers 2023, 104801Background
To transform the commodity wastes into high performance materials such as catalyst supports can facilitate the circular economy. Therefore, pitch-based carbon (MPC) which is mainly regarded as a premium carbon byproduct from the petroleum refinery was used to synthesize the supported amino catalyst for the Henry reaction of furfural.Methods
The amino functionalized pitch-based carbon was prepared by the facile chemical nitration and reduction. The surface textures of the functionalized carbon materials were carefully scrutinized by various characterization techniques, including PXRD, XPS, FT-IR, 13C SSNMR, and Raman spectroscopy. Meanwhile, the amount of surface accessible amino groups was quantified by the colorimetric method using 2-iminothiolane hydrochloride (ITL) as molecular probe.Significant finding
The results reveal that the surface amino groups were immobilized and quantified as 0.391 mmol/g at the periphery of MPC. Moreover, this amino functionalized carbon denoted NRE-MPC could catalyze the Henry reaction of furfural with nitromethane in isopropanol at 90 °C for 6 h to yield 2-(2-nitrovinyl)furan (NVF) as the major product in 65% with 87% furfural conversion. NRE-MPC can be also easily recycled at least four times and the peripheral amino groups alongside the graphitic domains of MPC were still chemically active with perceptible reactivity reduction.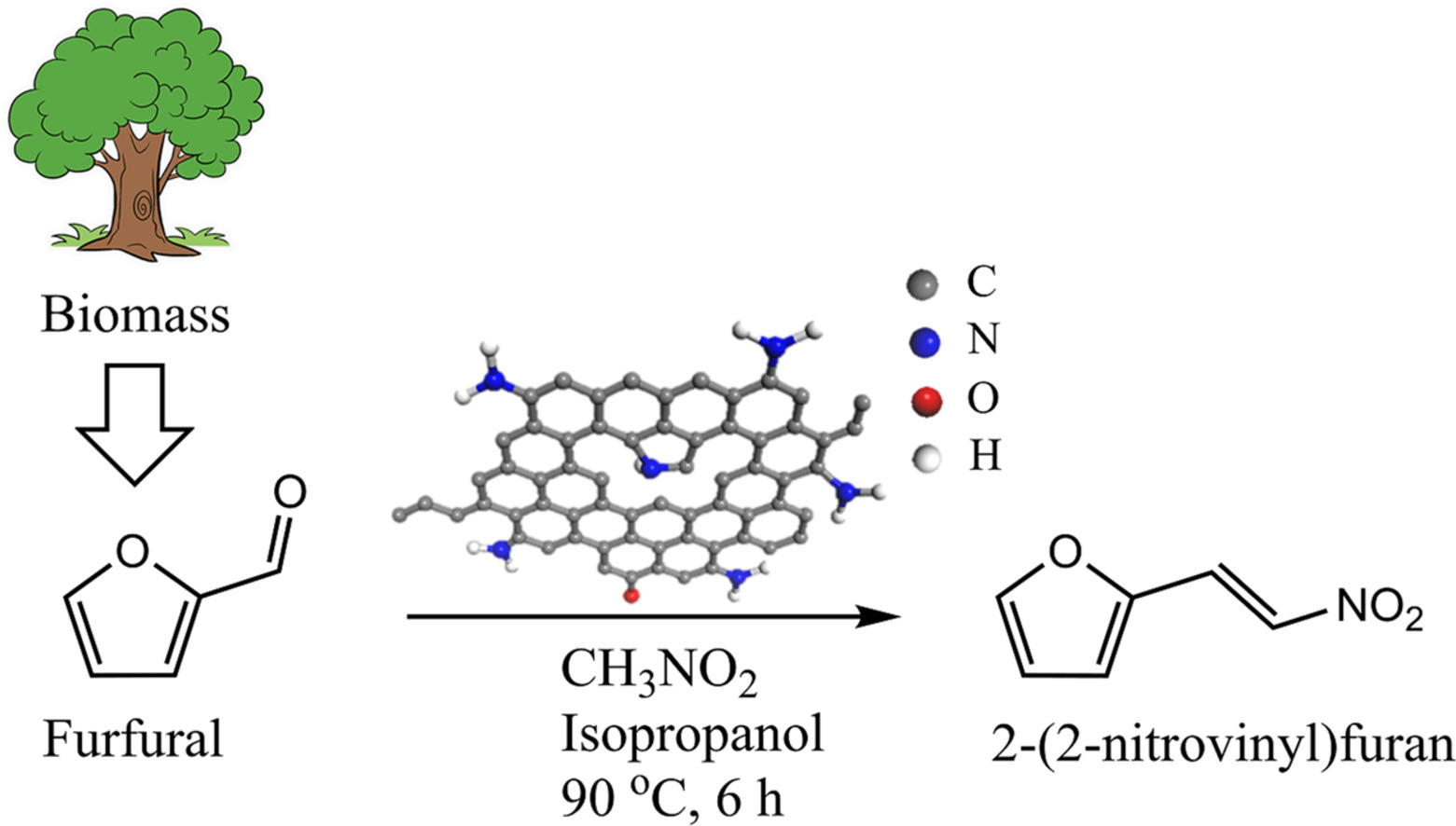
-
Facile synthesis of the atomically dispersed hydrotalcite oxide supported copper catalysts for the selective hydrogenation of 5–hydroxymethylfurfural into 2,5-bis(hydroxymethyl)furan
Raju Kumar, Hsin-Hui Lee, En Chen, Yuan-Peng Du, Chan-Yi Lin, Warot Prasanseang, Thanasak Solos, Kittisak Choojun, Tawan Sooknoi, Rui-Kun Xie, Jyh-Fu Lee, Po-Wen Chung*Applied Catalysis B: Environmental 2023, 329, 122547The selective hydrogenation of 5-hydroxymethylfurfural (HMF) to 2,5-bis(hydroxymethyl)furan (BHMF) using the atomically dispersed supported copper catalyst is investigated. The hydrotalcite oxide supported copper materials (Cu(x)HTO) are facilely prepared by coprecipitating metal precursors in a methanolic solution under a tuned pH. The surface characterization involving PXRD, TEM, H2/N2O-TPR, and XAS reveals unequivocal evidence for the presence of the atomically dispersed copper on HTO surface. XAS specifically indicates the formation of mononuclear copper species, and H2/N2O-TPR strongly supports the copper atoms of Cu(5)HTO are evenly distributed in 99% dispersion. Moreover, the reduced Cu(5)HTO (r-Cu(5)HTO) enables to completely hydrogenate HMF to BHMF under mild conditions, in comparison to the poor reactivity catalyzed by the hydrotalcite oxide supported copper nanoparticles (r-Cu(4)@HTO). The dramatic enhancement of HMF hydrogenation catalyzed by r-Cu(5)HTO can be attributed to the fine distribution of copper atoms which are situated homogeneously over HTO surface as well as chemically reactive for the carbonyl group.
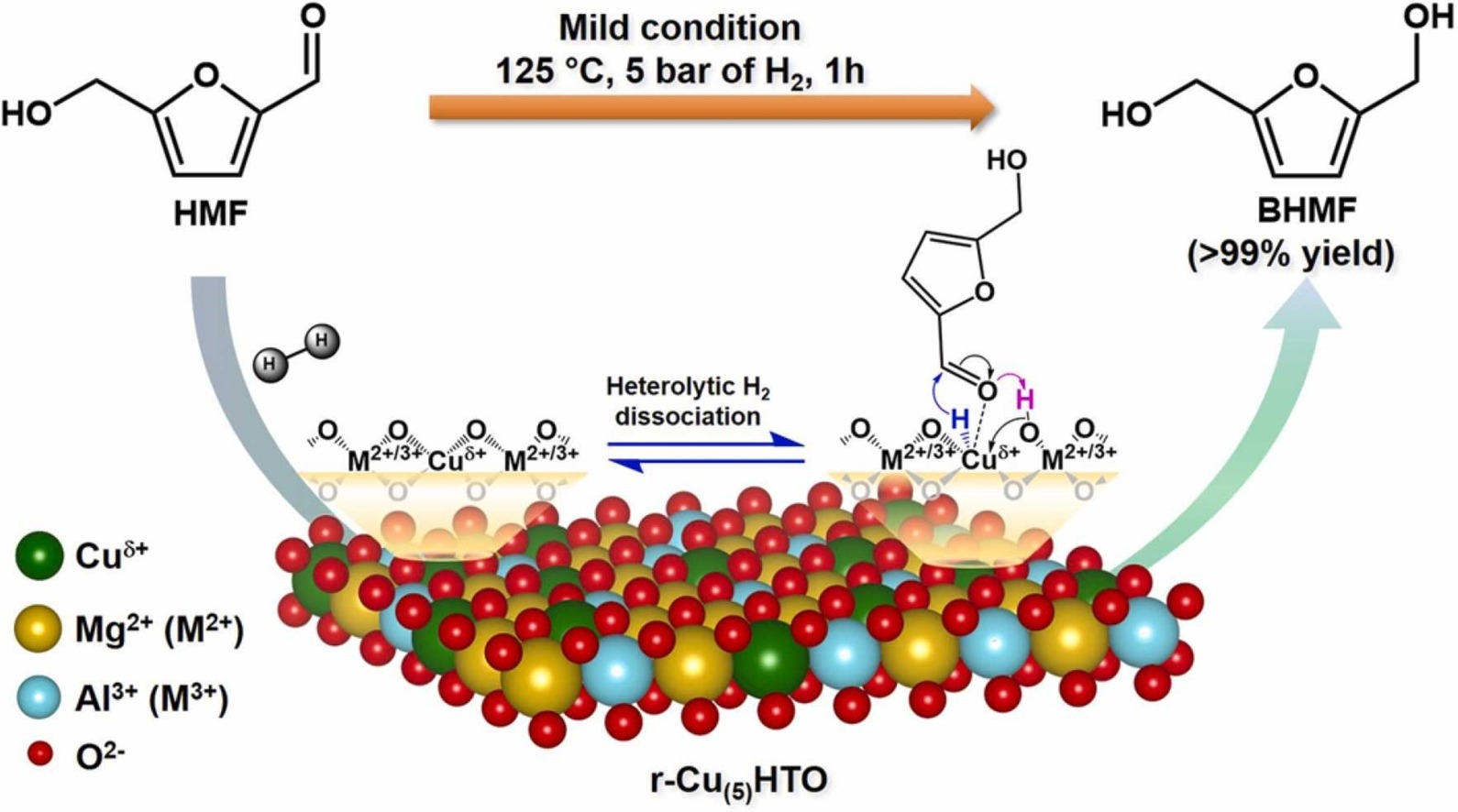
-
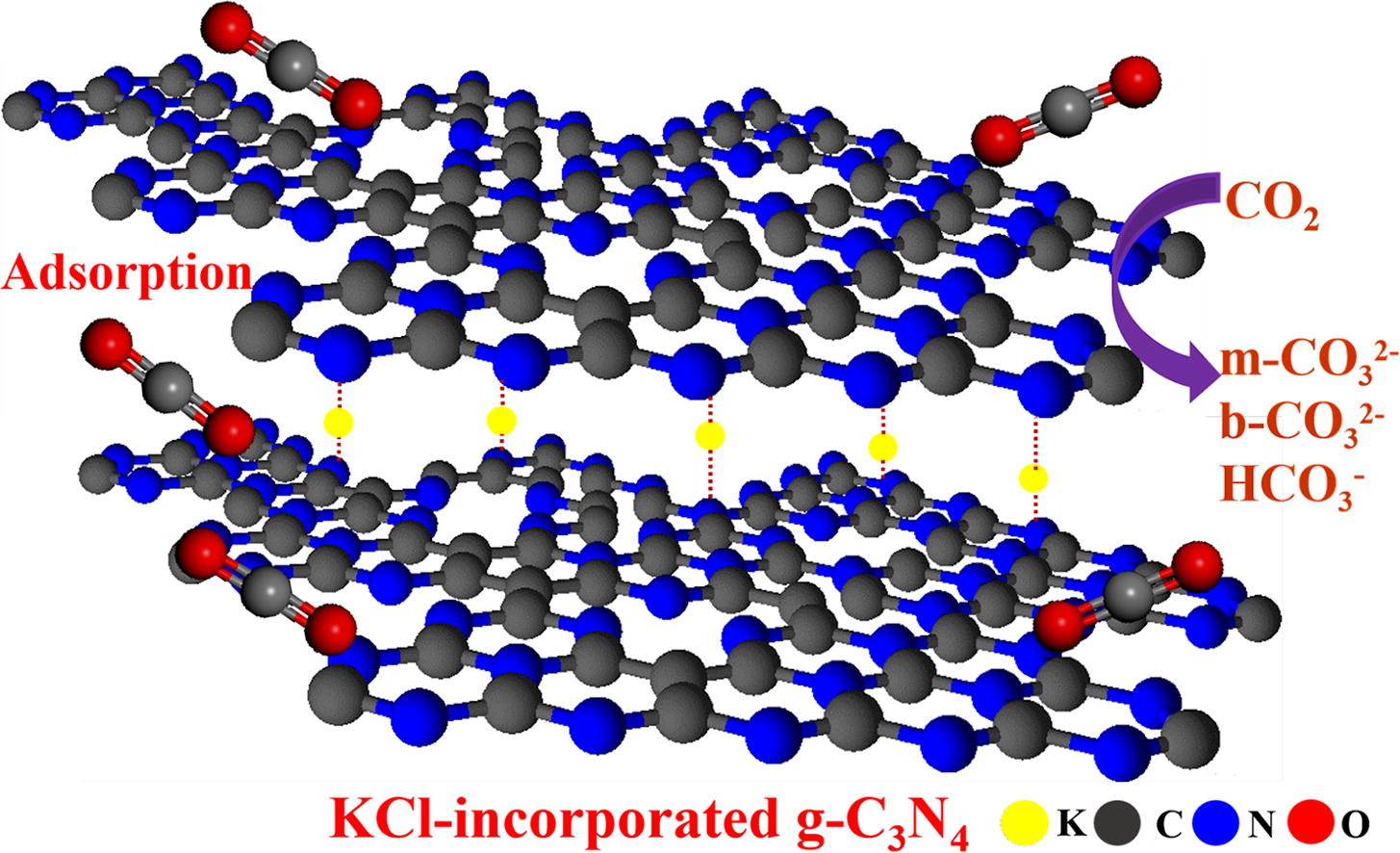
Understanding the intermediates and carbon dioxide adsorption of potassium chloride-incorporated graphitic carbon nitride with tailoring melamine and urea as precursors
Hepsiba Niruba Catherine, Zhi-Ting Liu, Chan-Yi Lin, Po-Wen Chung, Shun Tsunekawa, Shawn D. Lin, Masaaki Yoshida, Chechia Hu*Journal of Colloid and Interface Science 2023, 633, 598In this study, we demonstrated the synthesis of potassium chloride (KCl)-incorporated graphitic carbon nitride, (g-C3N4, CN) with varying amounts of N-vacancies and pyridinic-N as well as enhanced Lewis basicity, via a single-step thermal polymerization by tailoring the precursors of melamine and urea for carbon oxide (CO2) capture. Melamine, as a precursor, undergoes a phase transformation into melam and triazine-rich g-C3N4, whereas the addition of urea polymerizes the mixture to form melem and heptazine-rich g-C3N4 (CN11). Owing to the abundance of pyridinic-N and the high surface area, CN11 adsorbed higher amounts of CO2 (44.52 μmol m−2 at 25 °C and 1 bar of CO2) than those reported for other template-free carbon materials. Spectroscopic analysis revealed that the enhanced CO2 adsorption is due to the presence of pyridinic-N and Lewis basic sites on the surface. The intermediates of CO2 adsorption, including carbonate and bicarbonate species, attached to the CN samples were identified using in-situ Fourier-transform infrared spectroscopy (FTIR). This work provides insights into the mechanism of CO2 adsorption by comparing the structural features of the synthesized KCl-incorporated g-C3N4 samples. CN11, with an excellent CO2 uptake capacity, is viewed as a promising candidate for CO2 capture and storage.
2022
-
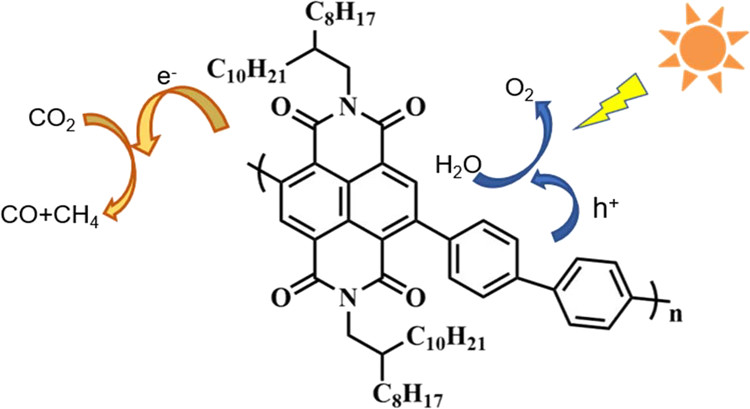
Naphthalene diimide-based donor–acceptor–donor small molecules as metal-free organocatalysts for photocatalytic CO2 reaction
Shih-Hao Wang, Pin-Zhen Chen, Yen-Yu Chen, Farheen Khurshid, Cheng-Wei Cai, Yu-Ying Lai*, Po-Wen Chung, Ru-Jong Jeng*, Syang-Peng Rwei, Leeyih Wang*ACS Applied Materials & Interfaces 2022, 14, 43109The application of organic small molecules as metal-free photocatalysts for light-driven photoreduction of carbon dioxide (CO2) has seldom been explored. This work developed four naphthalene diimide (NDI)-derived donor–acceptor–donor small molecules with different numbers of thiophene units, namely, NDI-2T, NDI-TT, NDI-4T, and NDI-6T, as metal-free photocatalysts to catalyze the reduction of CO2 under irradiation with an air mass 1.5G solar simulator at one-sun intensity. The structure–property relationship was investigated by exploring the effects of the electron-donating ability of the donor units on the optical properties, redox potential, electron–hole distribution, and exciton lifetime. NDI-6T exhibited the most red-shifted absorption, longest exciton lifetime, and strongest electron–hole separation. However, the large upshift in oxidation potential because of the elevated electron-donating ability of the hexathiophene unit significantly reduced the driving force for catalyst regeneration, leading to poor catalytic performance. Alternatively, NDI-4T possessed proper redox potentials, reduced charge-transfer resistance, and excellent photocurrent intensity; therefore, it effectively converted CO2 to a single product of CO in the presence of water as an electron donor without a sacrificial reagent or cocatalyst with a product yield of 168.6 μmol gcat–1 24 h–1, which was considerably higher than those of NDI-TT (111.9 μmol gcat–1 24 h–1), NDI-2T (88.4 μmol gcat–1 24 h–1), and NDI-6T (40.5 μmol gcat–1 24 h–1). This study provides a practical guideline for the molecular design of conjugated organic molecules as promising photocatalysts for CO2 photoreduction.
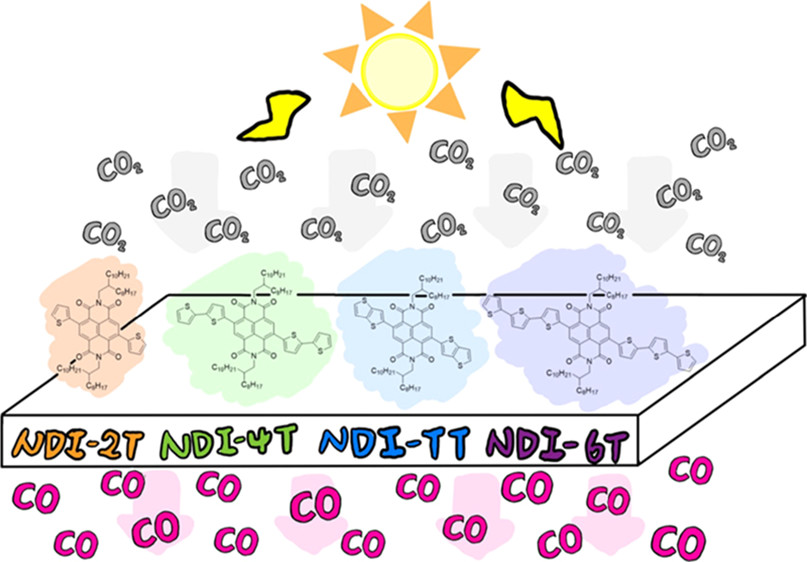
-
Silica-supported nanoscale hydrotalcite-derived oxides for C4 Chemicals from ethanol condensation
Raju Kumar, Cheng-Chia Li, Chia-Hui Wu, Tai-Wei Tzeng, Der-Lii M Tzou, Yu-Chuan Lin*, Po-Wen Chung*ACS Applied Nano Materials 2022, 5, 7885We demonstrated that the surface nature of nanoscale hydrotalcite-derived oxide (HTO) can be simply tailored through the addition of amorphous silica during the coprecipitation process of material preparation. The change in physical/chemical properties of the modified surface has been carefully scrutinized by a series of surface characterization techniques. The results showed that the HTO nanocomposite has molecular interactions with amorphous silica to form ternary Mg, Al, and Si interfaces, in accordance with enhancing chemically accessible aluminum (Al3+tetra) as acidic sites. Moreover, the silica-supported HTO (HTO/SiO2-X) could catalyze ethanol condensation to foster C4 chemicals at 250 °C through the flow reactor. HTO with a strong basic nature promoted 1-butanol production (66% selectivity), while HTO/SiO2-5 with more well-distributed acidic sites favored 1,3-butadiene production (43% selectivity). This difference in catalytic performance could be attributed to the redistribution of acidic and basic sites upon the newly formed ternary interfaces, which could facilitate the surface-mediated Meerwein–Ponndorf–Verley (MPV) reduction reaction due to the relatively high ethanol affinity.
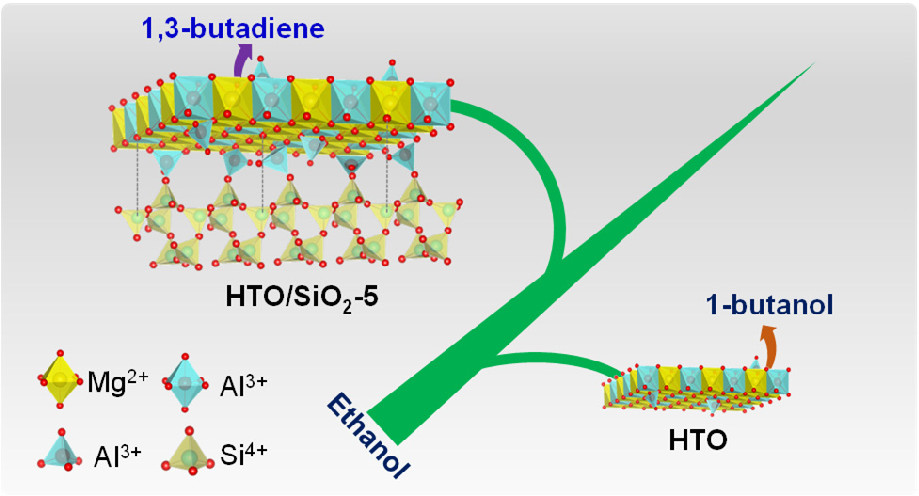
-
Solution-processable naphthalene diimide-Based conjugated polymers as organocatalysts for photocatalytic CO2 reaction with extremely stable catalytic activity for over 330 hours
Shih-Hao Wang, Farheen Khurshid, Pin-Zhen Chen, Ying-Ren Lai, Cheng-Wei Cai, Po-Wen Chung, Michitoshi Hayashi*, Ru-Jong Jeng*, Syang-Peng Rwei, Leeyih Wang*Chemistry of Materials 2022, 34, 4955Nowadays, the photochemical conversion of CO2 to high-value products is attracting tremendous research interest. Developing artificial photocatalysts with excellent catalytic activity and long-term stability is still a challenge. This work demonstrates that solution-processable naphthalenedimide (NDI)-based conjugated polymers, PNDI-BT, PNDI-DTBT, and PNDI-BP, which are copolymerization products of NDI with bithiophene (BT), 4,7-di(thiophen-2-yl)benzo[c][1,2,5]thiadiazole (DTBT), and biphenyl (BP), respectively, were capable of catalyzing the photochemical reduction of CO2 to produce CO in the presence of water without the need for metal-containing co-catalysts or sacrificial agents. In particular, the PNDI-BP-catalyzed reaction generated CH4 as well as CO. Results from time-resolved photoluminescence, photovoltage decay, electrochemical impedance spectroscopy, and transient photocurrent response experiments indicate that PNDI-BP with the largest dihedral angles along the conjugated backbone possessed the longest electron lifetime, the lowest charge-carrier recombination rate, and the smallest interfacial charge transfer resistance. Consequently, it had the best catalytic performance. Notably, PNDI-BP exhibited excellent recyclability, robust structural stability, and extremely steady catalytic activity for more than 330 h during a photocatalytic CO2 reaction. Furthermore, the solution-processability of the linear polymer allows the incorporation of porous substrates, which improve the reaction interface. The catalyst system of PNDI-BP@molecular sieves with H2O/triethylamine doubled the CO yield to 214.8 μmol·gcat–1 and enhanced the CH4 yield by ∼36 times to 61.4 μmol·gcat–1 in an 18 h reaction.
-
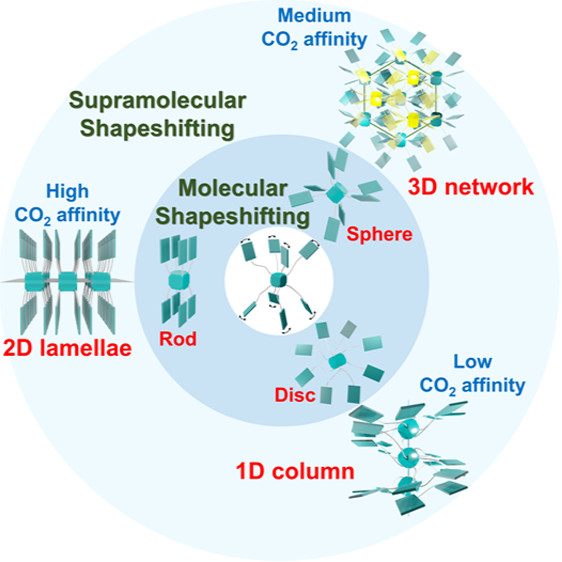
Connecting molecular and supramolecular shapeshifting by the Ostwald’s nucleation stages of a star giant molecule
Hong-Yu Hsu, Jou-Tsen Ou, Tsu-Tung Cheng, Heng-Yi Lin, Chan-Yi Lin, Jia Chen, Bin Sun, Po-Wen Chung*, Chien-Lung Wang*Journal of the American Chemical Society 2022, 144The shapeshifting behavior for synthetic matters was found at either the molecular or supramolecular level, but the connection between shapeshifting at the two hierarchical levels remains missing. In this study, an 8-arm star giant molecule, NPOSS, was synthesized to connect the molecular and supramolecular shapeshifting. Controlling the conditions of bulk self-assembly allowed us to bring NPOSS into three different Ostwald’s stages of nucleation. The high conformational flexibility of NPOSS facilitates molecular shapeshifting and allows NPOSS to take the discotic, rod-like and star-like geometries in different Ostwald’s stages. Simultaneous changes in the supramolecular scaffolds were observed as the discotic, rod-like and star-like NPOSS molecules self-assembled into the supramolecular scaffolds of 1D columns, 2D lamellae, and 3D networks, respectively. These changes in the hierarchical structures also affect the CO2 affinity of NPOSS. Therefore, the connection between the molecular/supramolecular shapeshifting and the structure-driven property changes of NPOSS were established by taking advantage of the high conformational freedom of the 8-arm star giant molecule and its diverse self-assembly pathways leading to the different Ostwald’s stages.
-
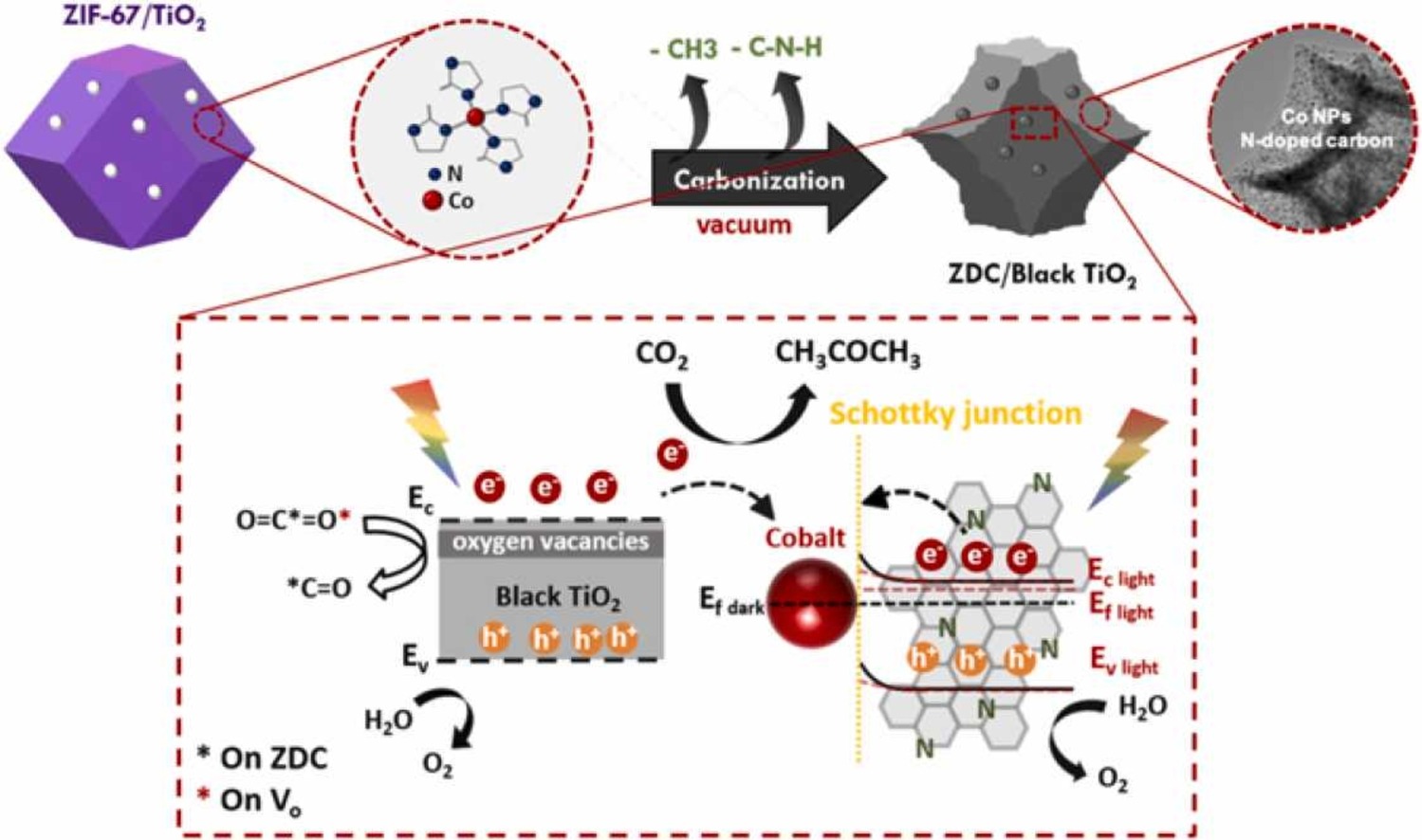
Photocatalytic CO2 reduction for C2-C3 oxy-compounds on ZIF-67 derived carbon with TiO2
Pei-Hsun Sung, Chih-Yang Huang, Chan-Yi Lin, Po-Wen Chung, Yu-Chung Chang, Li-Chyong Chen, Hsin-Yu Chen, Chien-Neng Liao*, Erh-Li Chiu, Cheng-Yu Wang*Journal of CO2 Utilization 2022, 58, 101920Excess carbon dioxide emission accompanied by gross fossil fuel consumption has worsened the global warming crisis in recent decades. Photo-induced conversion of CO2 into hydrocarbons is a potential solution to mitigate both energy and environmental problems. However, conventional photocatalysts suffer low efficiency in CO2 conversion with often C1 and/or C2 products. In this work, pyrolyzed cobalt type zeolitic imidazolate framework (ZIF-67), namely ZIF-derived carbon (ZDC) with residual Co nanoparticles and the corresponding ZDC/TiO2 composite (ZDC/T), were adopted as gas-phase CO2 reduction photocatalysts. The ZDC photocatalyst is inclined to form C2 compound (acetaldehyde), while the ZDC/T composite has high selectivity in C3 product (acetone). High pyrolysis temperature would degrade the photocatalytic performance in neat ZDC due to enhanced cobalt aggregation and nitrogen loss after breaking ZIF-67 Co-N coordination. Alternatively, the pyrolyzed ZDC/T revealed superior photocatalytic CO2 reduction performance because the Co-N bonds were found to decrease without changing Co nanoparticle size and dissociation of nitrogen notably. In conclusion, we synthesized novel photocatalysts for multi-carbon (C2-C3) products with high selectivity, and provided a plausible mechanism to illustrate the formation process of multi-carbon products during photoreduction of CO2.
-
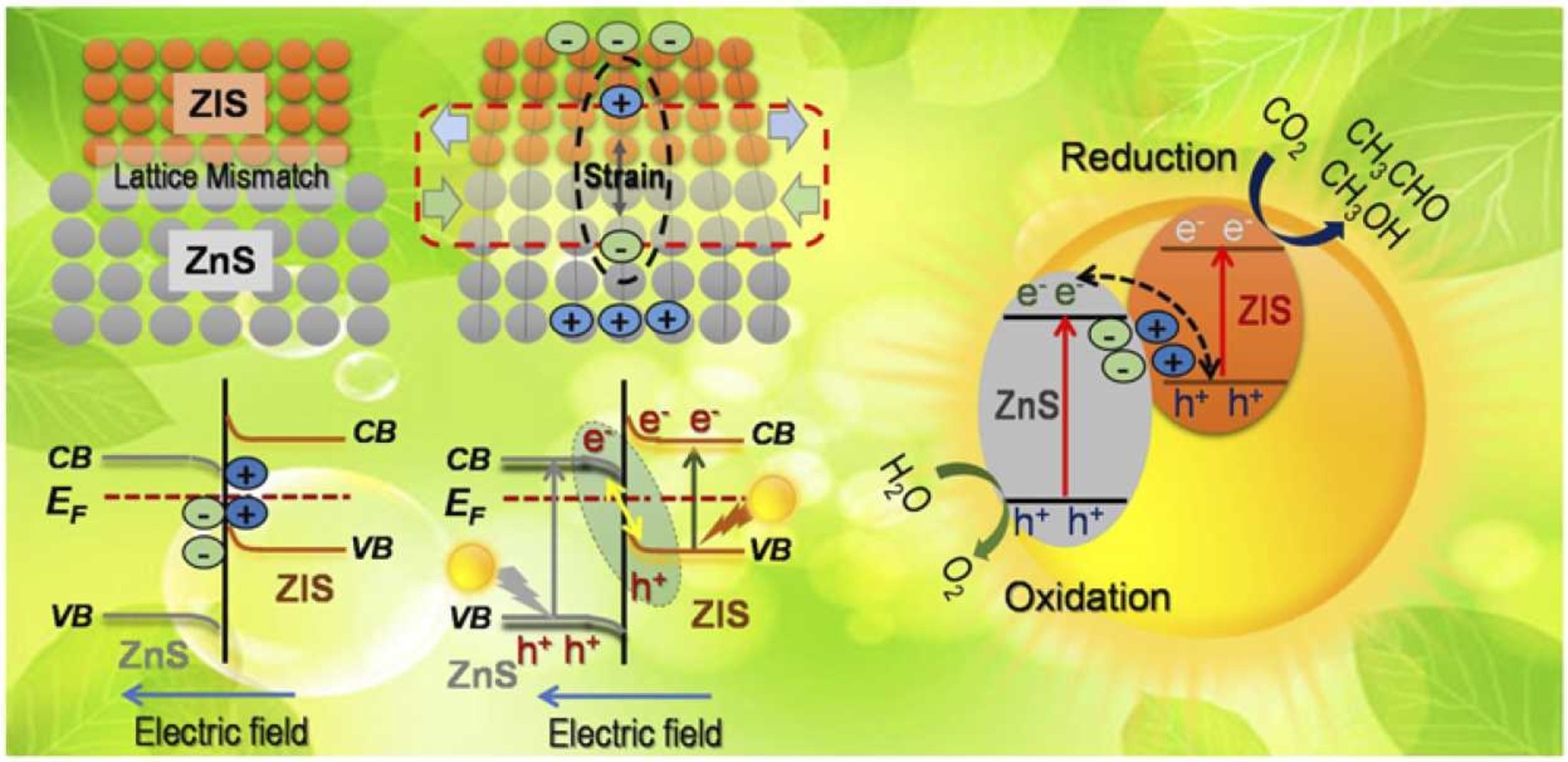
Boosting photocatalytic CO2 reduction in a ZnS/ZnIn2S4 heterostructure through strain-induced direct Z-scheme and a mechanistic study of molecular CO2 interaction thereon
Amr Sabbah, Indrajit Shown, Mohammad Qorbani, Fang-Yu Fu, Tsai-Yu Lin, Heng-Liang Wu, Po-Wen Chung, Chih-I Wu, Svette Reina Merden Santiago, Ji-Lin Shen, Kuei-Hsien Chen*, Li-Chyong Chen*Nano Energy 2022, 93, 106809Employing direct Z-scheme semiconductor heterostructures in photocatalysis offers efficient charge carrier separation and isolation of both redox reactions, thus beneficial to reduce CO2 into solar fuels. Here, a ZnS/ZnIn2S4 heterostructure, comprising cubic ZnS nanocrystals on hexagonal ZnIn2S4 (ZIS) nanosheets, is successfully fabricated in a single-pot hydrothermal approach. The composite ZnS/ZnIn2S4 exhibits microstrain at its interface with an electric field favorable for Z-scheme. At an optimum ratio of Zn:In (~ 1:0.5), an excellent photochemical quantum efficiency of around 0.8% is reached, nearly 200-fold boost compared with pristine ZnS. Electronic levels and band alignments are deduced from ultraviolet photoemission spectroscopy and UV-Vis. Evidence of the direct Z-scheme and carrier dynamics is verified by photo-reduction experiment, along with photoluminescence (PL) and time-resolved PL. Finally, diffuse-reflectance infrared Fourier transformed spectroscopy explores the CO2 and related intermediate species adsorbed on the catalyst during the photocatalytic reaction. This microstrain-induced direct Z-scheme approach opens a new pathway for developing next-generation photocatalysts for CO2 reduction.
-
Metal-free four-in-one modification of g-C3N4 for superior photocatalytic CO2 reduction and H2 evolution
Mahmoud Kamal Hussien, Amr Sabbah, Mohammad Qorbani, Mohamed Hammad Elsayed, Putikam Raghunath, Tsai-Yu Lin, Shaham Quadir, Hong-Yi Wang, Heng-Liang Wu, Der-Lii M Tzou, Ming-Chang Lin, Po-Wen Chung, Ho-Hsiu Chou, Li-Chyong Chen*, Kuei-Hsien Chen*Chemical Engineering Journal 2022, 430, 132853Utilization of g-C3N4 as a single photocatalyst material without combination with other semiconductor remains challenging. Herein, we report a facile green method for synthesizing a metal free modified g-C3N4 photocatalyst. The modification process combines four different strategies in a one-pot thermal reaction: non-metal doping, porosity generation, functionalization with amino groups, and thermal oxidation etching. The as-prepared amino-functionalized ultrathin nanoporous boron-doped g-C3N4 exhibited a high specific surface area of 143.2 m2 g−1 which resulted in abundant adsorption sites for CO2 and water molecules. The surface amino groups act as Lewis basic sites to adsorb acidic CO2 molecules, which can also serve as active sites to facilitate hydrogen generation. Besides, the simultaneous use of ammonium chloride as a dynamic gas bubble template along with thermal oxidation etching efficiently boosts the delamination of the g-C3N4 layers to produce ultrathin sheets; this leads to stronger light–matter interactions and efficient charge generation. Consequently, the newly modified g-C3N4 achieved selective gas-phase CO2 reduction into CO with a production yield of 21.95 µmol g−1, in the absence of any cocatalyst. Moreover, a high hydrogen generation rate of 3800 µmol g-1h−1 and prominent apparent quantum yield of 10.6% were recorded. This work opens up a new avenue to explore different rational modifications of g-C3N4 nanosheets for the efficient production of clean energy.
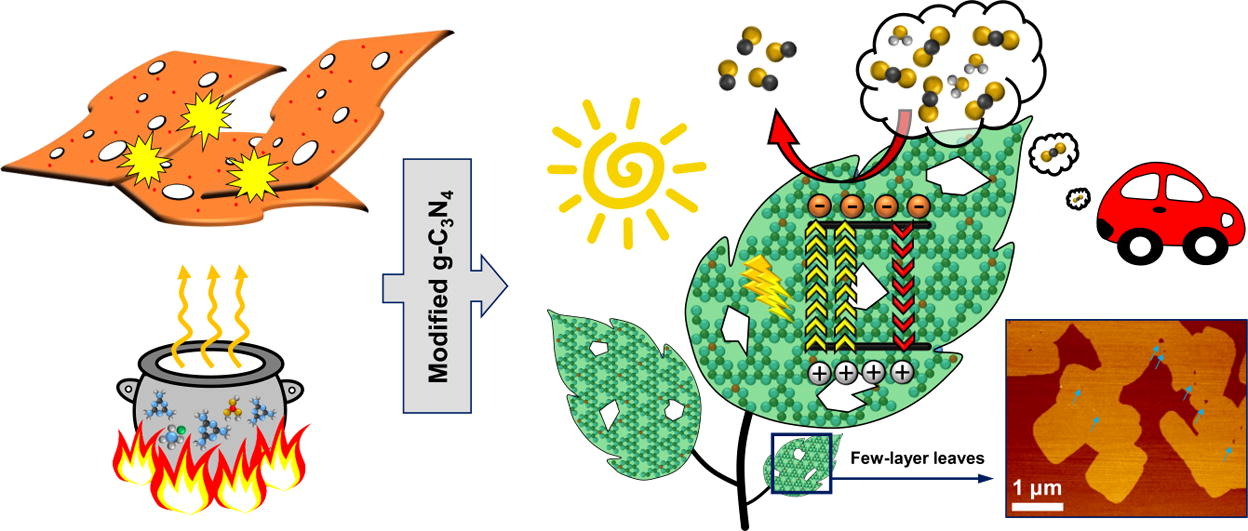
A simple and environmentally benign one-pot thermal process has been employed to synthesize a novel amine-functionalized ultrathin nanoporous boron-doped g-C3N4. The modified g-C3N4 acts as a porous low-dimensional artificial leaf with excellent photocatalytic activity toward CO2 reduction and H2 generation. -
Chromatic fulleropyrrolidine as long‐lived metal‐free catalyst for CO2 photoreduction reaction
Shih-Hao Wang, Rathinam Raja, Chuen-Yo Hsiow, Farheen Khurshid, Hau-Ren Yang, Po-Wen Chung, Yu-Ying Lai, Ru-Jong Jeng*, Leeyih Wang*ChemSusChem 2022, 15, e202102476.Conversion of CO2 into carbonaceous fuels with the aid of solar energy has been an important research subject for decades. Owing to their excellent electron-accepting capacities, fullerene derivatives have been extensively used as n-type semiconductors. This work reports that the fulleropyrrolidine functionalized with 4,7-di(thiophen-2-yl)benzo[c][1,2,5]thiadiazole, abbreviated as DTBT-C60, could efficiently catalyze the photoreduction of CO2 to CO. The novel C60-chromophore dyad structure facilitated better usage of solar light and effective dissociation of excitons. Consequently, the DTBT-C60 exhibited a promising CO yield of 144 μmol gcat−1 under AM1.5G solar illumination for 24 h. Moreover, the isotope experiments demonstrated that water molecules could function as an electron source to reactivate DTBT-C60. Impressively, DTBT-C60 exhibited an extremely durable catalytic activity for more than one week, facilitating the practical application of photochemical CO2 reaction.
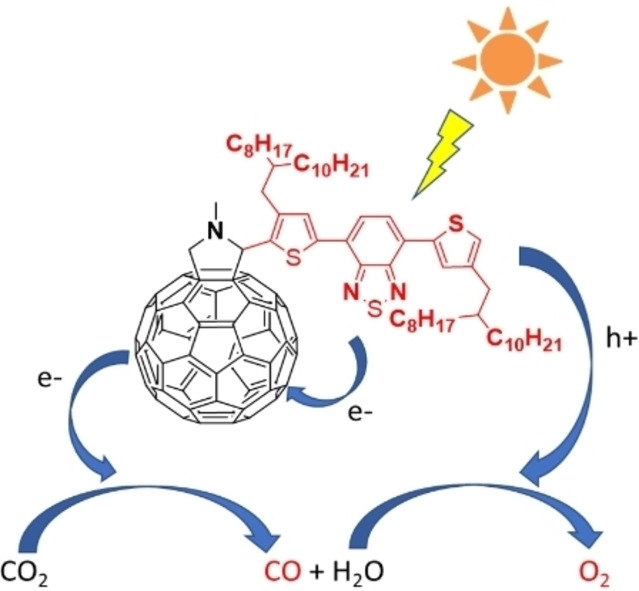
Molecular photocatalyst: C60-chromophore dyad effectively catalyzes the photoreduction reaction of CO2 to produce CO with 100 % selectivity by directly using water molecules as a source of electrons to regenerate chromophore units. The dyad structure markedly enhances the dissociation efficiency of excitons and reduces the recombination rate of charge carriers. This molecular catalyst exhibits a steady catalytic activity during a 7-day photoreaction.
2021
-
Tai materials engineering of violin soundboards by stradivari and guarneri
Cheng-Kuan Su, Szu-Yu Chen, Jen-Hsuan Chung, Guo-Chian Li, Brigitte Brandmair, Thomas Huthwelker, John L Fulton, Camelia N Borca, Shing-Jong Huang, Joseph Nagyvary, Hsiao-Han Tseng, Chih-Hui Chang, Dai-Ting Chung, Rafael Vescovi, Yi-Shiuan Tsai, Wenjie Cai, Bing-Jyun Lu, Jia-Wei Xu, Chia-Shuo Hsu, Jun-Jie Wu, Hao-Zhi Li, Yu-Kai Jheng, Sheng-Fong Lo, Hao Ming Chen, Yi-Ting Hsieh, Po-Wen Chung, Chien-Sheng Chen, Yuh-Chang Sun, Jerry Chun Chung Chan, Hwan-Ching Tai*Angewandte Chemie International Edition 2021, 60, 19144We investigated the material properties of Cremonese soundboards using a wide range of spectroscopic, microscopic, and chemical techniques. We found similar types of spruce in Cremonese soundboards as in modern instruments, but Cremonese spruces exhibit unnatural elemental compositions and oxidation patterns that suggest artificial manipulation. Combining analytical data and historical information, we may deduce the minerals being added and their potential functions—borax and metal sulfates for fungal suppression, table salt for moisture control, alum for molecular crosslinking, and potash or quicklime for alkaline treatment. The overall purpose may have been wood preservation or acoustic tuning. Hemicellulose fragmentation and altered cellulose nanostructures are observed in heavily treated Stradivari specimens, which show diminished second-harmonic generation signals. Guarneri's practice of crosslinking wood fibers via aluminum coordination may also affect mechanical and acoustic properties. Our data suggest that old masters undertook materials engineering experiments to produce soundboards with unique properties.
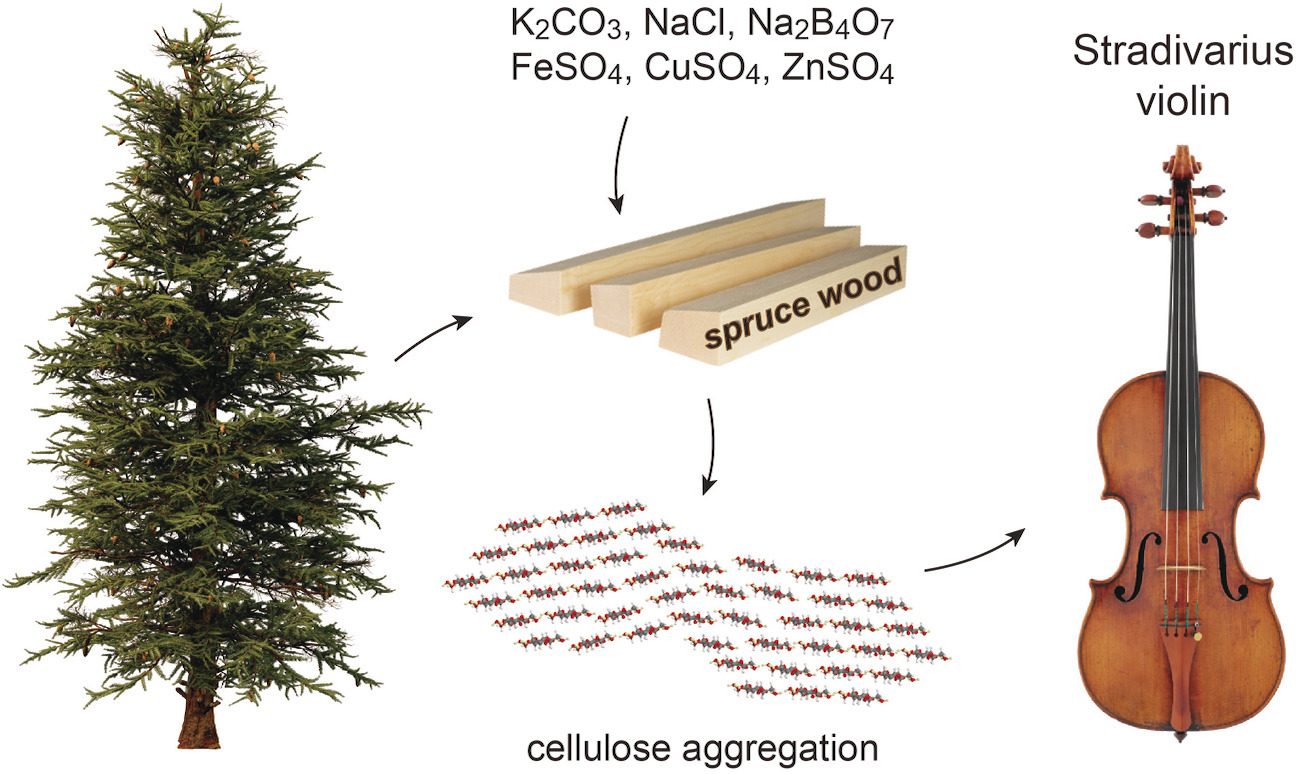
ICP-MS is used to identify the unique mineral recipe used by Antonio Stradivari to treat his spruce wood, the material for the violin top plate. Chemical manipulation led to hemicellulose fragmentation and cellulose rearrangement. Incorporating engineered wood may contribute to the unique tonal qualities of these acclaimed instruments. -
化工技術於友善農業應用–生質化學品發展的回顧與展望
Lin, C.-H. (林展逸); Chang, C.-C.(張志成); Chung, P.-W. (鍾博文)*工業材料雜誌 2021, 414, 74為達成二氧化碳淨零排放之願景,全球石化產業面臨尋求可替代性原物料與永續製程的挑戰,而從廢棄生物質衍生的生質化學品產業隸屬再生資源,如可漸進式導入石化產業鏈,提供產品多元選擇,則應可減緩消耗石化原料及實踐循環經濟宗旨。本文回顧國內外案例,闡述現今呋喃相關生質化學品開發之現況,並展望生物質應用於呋喃相關生質化學品之發展趨勢。
2020
-
Hydrophobic copper catalysts derived from copper phyllosilicates in the hydrogenation of levulinic acid to γ-valerolactone
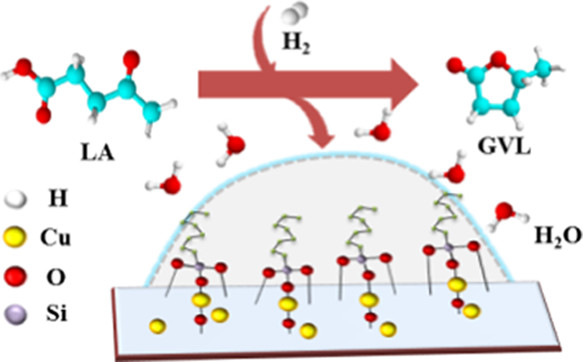
Ya-Ju Tsou, Thien Dien To, Yu-Chia Chiang, Jyh-Fu Lee, Raju Kumar, Po-Wen Chung, Yu-Chuan Lin*ACS Applied Materials & Interfaces 2020, 12 54851A reduction–silylation–reduction method was developed to synthesize hydrophobic Cu catalysts derived from Cu phyllosilicates (CuPS). Triethoxy(octyl)silane (OTS) was used as the coupling agent. The OTS-grafted, reduced CuPS catalysts were applied in the hydrogenation of levulinic acid (LA) to γ-valerolactone (GVL). The most promising catalyst was synthesized by reducing CuPS at a high temperature (350 °C for 3 h), followed by OTS grafting, and then by repeating the previous reduction step. High LA conversion (95.7%), GVL yield (85.2%), and stability (3 cycles with a 7.5% loss of initial activity) were obtained at a mild reaction condition (130 °C with a H2 pressure of 12 bar). A high reduction temperature not only leads to a low oxidation state of Cu species but also suppresses the formation of silylation-induced acids. Moreover, the intrinsic activity of a reduced CuPS catalyst was nearly intact after subjecting to silylation and the second reduction treatment.
-
Understanding catalytic hydrogenolysis of 5-hydroxymethylfurfural (HMF) to 2,5-dimethylfuran (DMF) using carbon supported Ru catalysts
Tai-Wei Tzeng, Chan-Yi Lin, Chih-Wen Pao, Jeng-Lung Chen, Po-Wen Chung*Fuel Processing Technology 2020, 199, 106225Herein, our study has demonstrated that microporous carbon supported Ru catalysts can selectively convert 5-hydroxymethylfurfural (HMF) to 2,5-dimethylfuran (DMF) in isopropanol through hydrogenolysis in a catalytic ratio of 2.47mol% under 5 bar of hydrogen at 125 °C for 1 h. DMF yield can reach 69.52% with complete HMF conversion. The effects of reaction temperature, hydrogen pressure, metal loading and reaction solvent for hydrogenolysis have been carefully investigated. In addition, to better understand the influence of the intrinsic nature of carbonaceous material, carbon materials possessing different surface texture including microporosity, mesoporosity and nonporous carbon black were used as catalyst supports, and structural properties have been comprehensively characterized by powder X-ray diffraction spectrometer (PXRD), transmission electron microscopy (TEM), nitrogen and CO2 sorption, thermogravimetric analysis (TGA), and X-ray absorption spectroscopy (XAS). We have concluded that the nature of the carbonaceous material affects the Ru nanoparticle formation upon thermal reduction. The atomic configuration of Ru nanoparticles on carbon support (i.e., metallic Ru or RuOx) would direct the catalytic pathway differently and could be attributed to the interaction between ruthenium and carbon surface.
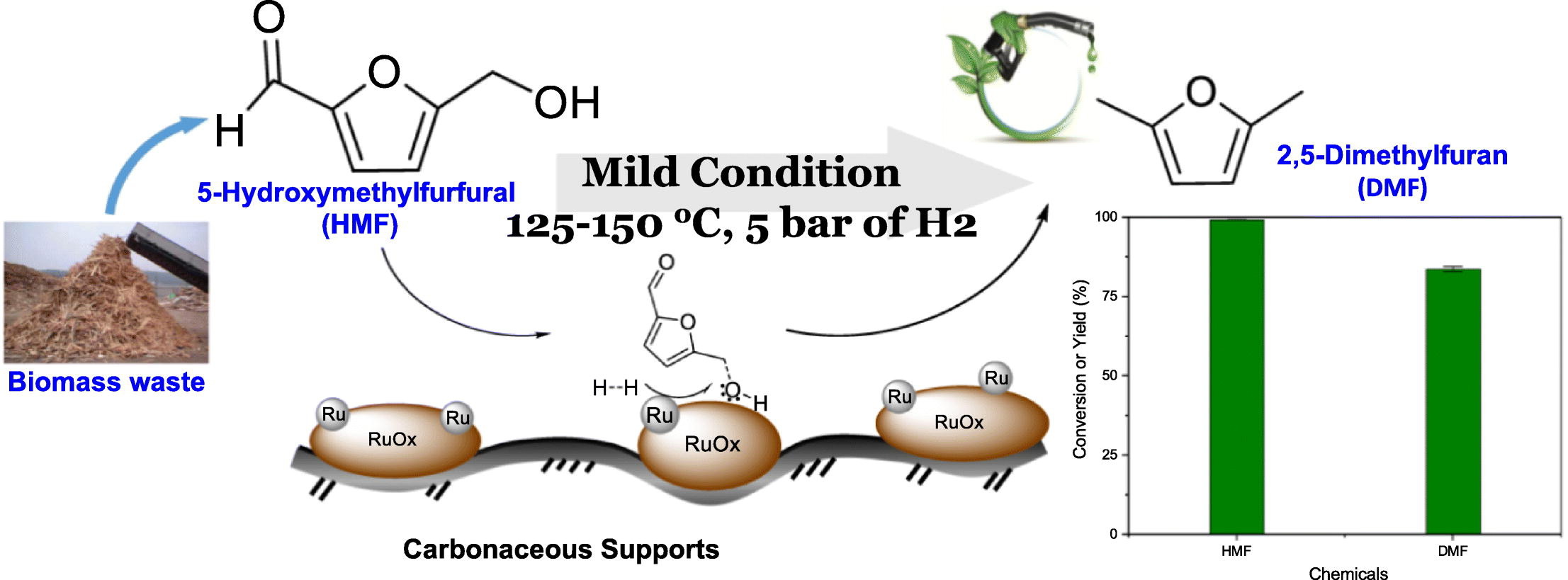
-
The oversolubility of methane gas in nano-confined water in nanoporous silica materials
Chih-Cheng Liu, Hao-Ju Chou, Chan-Yi Lin, Damodar Janmanchi, Po-Wen Chung, Chung-Yuan Mou, Steve S.-F. Yu, Sunney I. Chan*Microporous and Mesoporous Materials 2020, 293, 109793The oversolubility of the non-polar methane (CH4) gas in nano-confined liquid in nanoporous silica materials is investigated. A series of mesoporous silica materials with different pore sizes, pore volumes, and different amounts of nano-confined water (H2O) are prepared using a pore-expanding reagent and surfactants of different chain-lengths, and the CH4 absorption by the silica nano-materials is studied at 298 K under low CH4 gas pressures. Analysis of the CH4 absorption data reveals unequivocal evidence for oversolubility of CH4 in the nano-confined H2O of all the hydrated nano-materials. The solubility enhancements are several hundred fold relative to the CH4 solubility in bulk H2O. Interestingly, the enhancements are 25–30% higher when a tricopper cluster complex capable of efficient selective CH4 oxidation under ambient conditions is immobilized into the nano-channels of the silica materials. This dramatic enhancement of the CH4 solubility is attributed to specific CH4 binding to the tricopper cluster complexes embedded within the mesopores of the nanoporous materials.
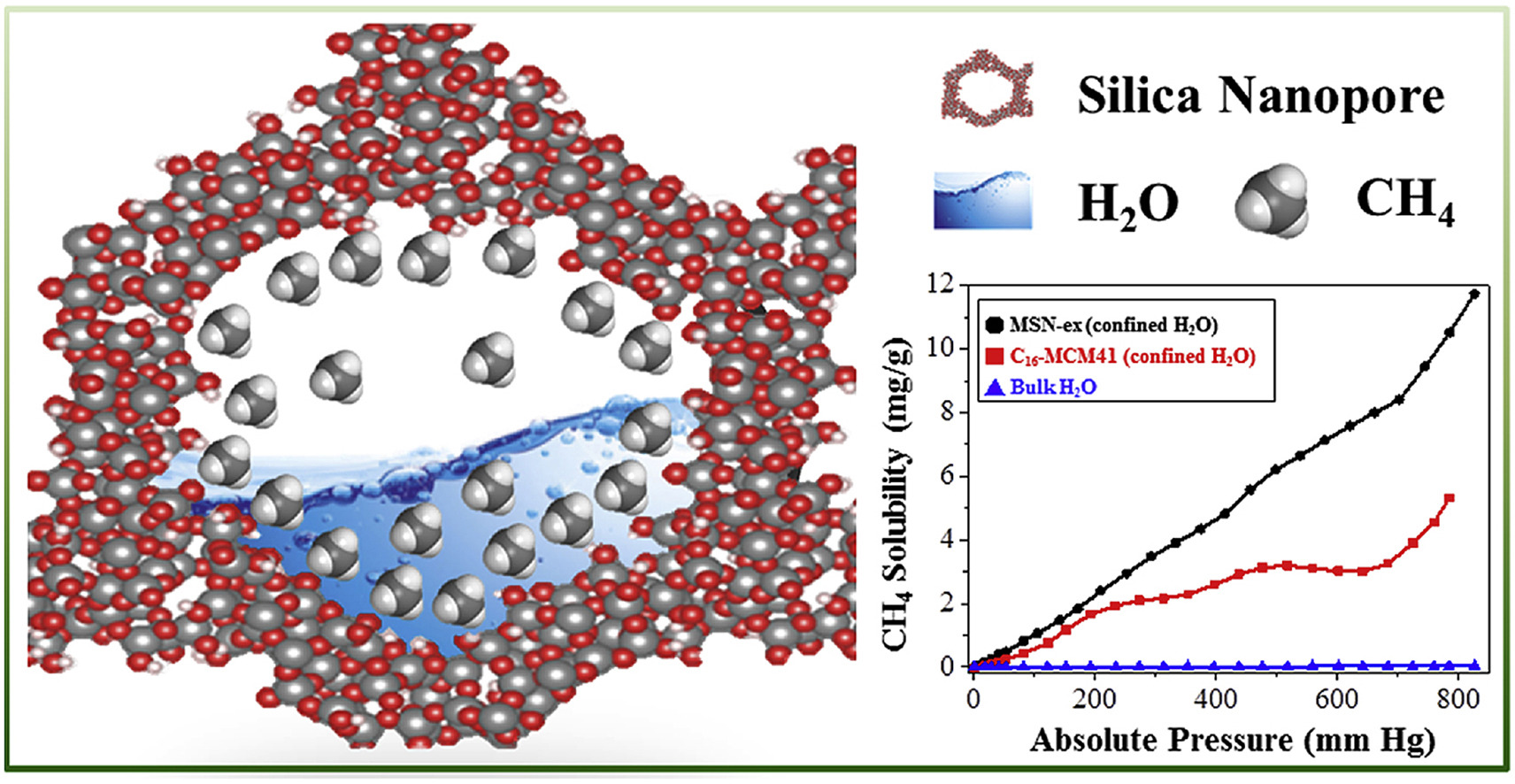
2019
-
Understanding the production of 5-hydroxymethylfurfural (HMF) from chitosan using solid acids
Tai-Wei Tzeng, Prasenjit Bhaumik, Po-Wen Chung*Molecular Catalysis 2019, 479, 110627Herein, we demonstrate that 5-hydroxymethylfurfural (HMF) can be chemically derived from commercial chitosan (CScom) using solid acids in a reaction carried out using Nafion®50 resin (NR50) in a co-solvent system of methyl isobutyl ketone (MIBK) and DI water (V/V = 2) at 180 °C for 2 h. After reaction, the products can be well resolved by HPLC and GC-FID and identified by GC–MS. HMF yields could reach up to 32.6 ± 4.2 mol %, which is comparable to HMF yields obtained by using homogeneous organic/inorganic acids. We surmise that interactions between the chitosan chains and the perfluorinated backbone of NR50 could interrupt the hydrogen-bonding network within the chitosan to facilitate hydrolysis and further dehydration. In addition, to understand the interactions between the chitosan monomers/oligomers with the surface of NR50 resin, gel permeation chromatography (GPC) is employed to monitor the evolution of oligomer distribution and HMF production after the reaction. The results suggest that chitosan-derived monomers/oligomers might adsorb on NR50, and D-glucosamine might favorably dehydrated for HMF production once hydrolyzed from chitosan.
-
KSCN-induced interfacial dipole in black TiO2 for enhanced photocatalytic CO2 reduction
Fang-Yu Fu, Indrajit Shown, Chia-Shuo Li, Putikam Raghunath, Tsai-Yu Lin, Tadesse Billo, Heng-Liang Wu, Chih-I Wu, Po-Wen Chung, Ming-Chang Lin, Li-Chyong Chen*, Kuei-Hsien Chen*ACS Applied Material Interfaces 2019, 11, 25186Tuning the electronic band structure of black titania to improve photocatalytic performance through conventional band engineering methods has been challenging because of the defect-induced charge carrier and trapping sites. In this study, KSCN-modified hydrogenated nickel nanocluster-modified black TiO2 (SCN–H–Ni–TiO2) exhibits enhanced photocatalytic CO2 reduction due to the interfacial dipole effect. Upon combining the experimental and theoretical simulation approach, the presence of an electrostatic interfacial dipole associated with chemisorption of SCN has dramatic effects on the photocatalyst band structure in SCN–H–Ni–TiO2. An interfacial dipole possesses a more negative zeta potential shift of the isoelectric point from 5.20 to 3.20, which will accelerate the charge carrier separation and electron transfer process. Thiocyanate ion passivation on black TiO2 demonstrated an increased work function around 0.60 eV, which was induced by the interracial dipole effect. Overall, the SCN–H–Ni–TiO2 photocatalyst showed an enhanced CO2 reduction to solar fuel yield by 2.80 times higher than H–Ni–TiO2 and retained around 88% product formation yield after 40 h.
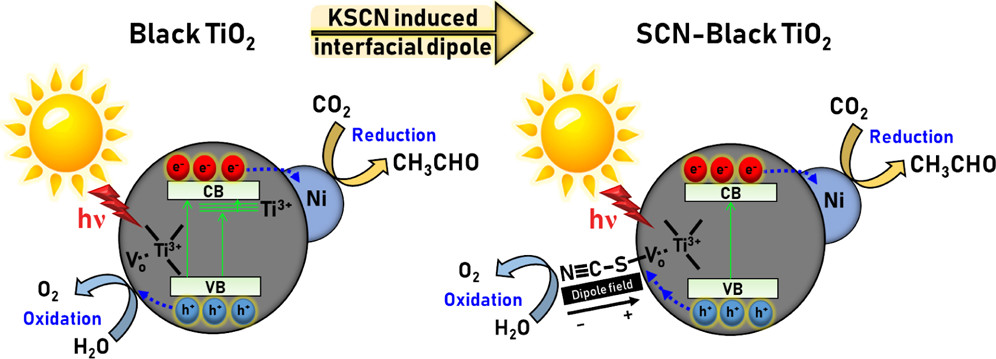
-
Unsupported and silica-supported perovskite-type lanthanum manganite and lanthanum ferrite in the conversion of ethanol
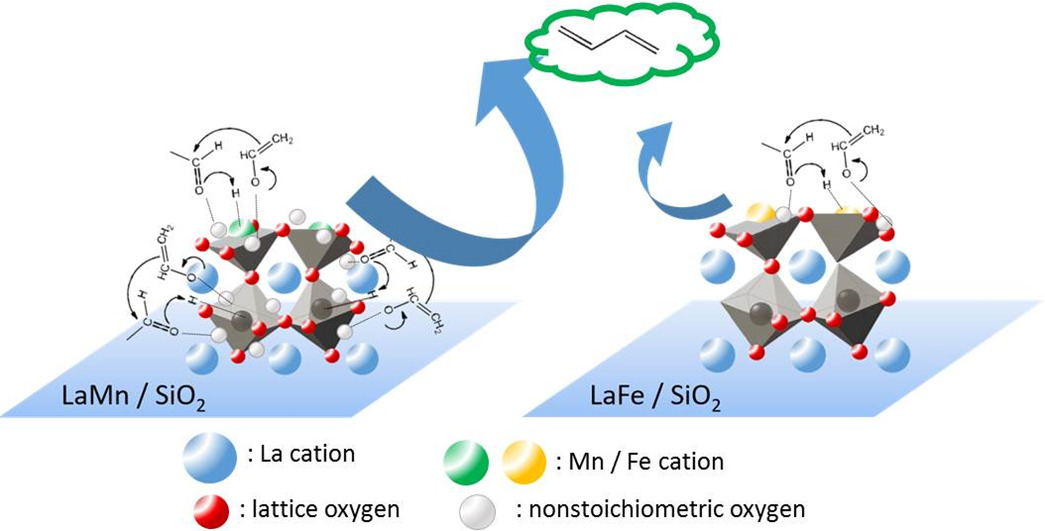
Ting-Fang Yu, Che-Wei Chang, Po-Wen Chung*, Yu-Chuan Lin*Fuel Processing Technology 2019, 194, 106117.The acid-base properties of perovskite-type LaMnO3 and LaFeO3 catalysts were adjustable by immobilizing them on silica. Unsupported LaMnO3 and LaFeO3 contained majorly basic sites on their surfaces. Dispersing LaMnO3 and LaFeO3 on silica suppressed their basicities and improved their acidities due to the increased concentrations of tetravalent B-site cations. Catalytic performances of ethanol conversion reflected the acid-base properties of bulk and silica-supported LaMnO3 and LaFeO3. Under differential analysis conditions, unsupported perovskites were active in base-catalyzed reactions such as reverse aldolization and Tishchenko reaction, while silica-supported perovskites were active in aldolization and dehydration. Furthermore, a higher aldolization activity of LaMnO3/SiO2 than that of LaFeO3/SiO2 was discovered. The different aldolization activities were attributed to excess mobile oxygen on the surface of LaMnO3/SiO2, forming aldolization-active Lewis acid (Mn4+)-base (nonstoichiometric oxygen) pairs. In contrast, the Lewis acid-base pair was absent in LaFeO3/SiO2, of which oxygen vacancies were enriched with. Oxygen nonstoichiometry was responsible for the varying aldolization activities of silica-supported LaMnO3 and LaFeO3 catalysts.
2018
-
The aldolization nature of Mn4+-nonstoichiometric oxygen pair sites of perovskite-type LaMnO3 in the conversion of ethanol
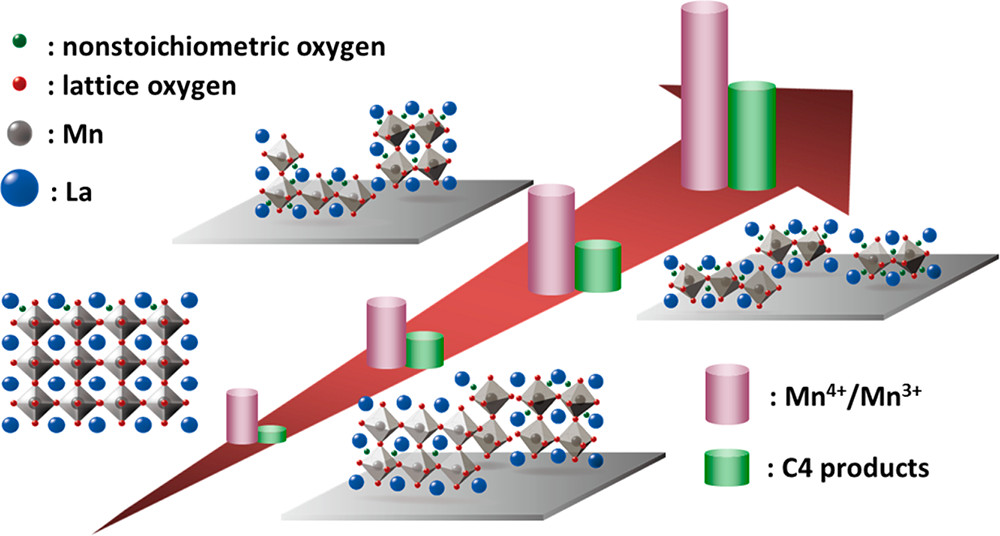
Ren-Kai Chen, Ting-Fang Yu, Meng-Xun Wu, Tai-Wei Tzeng, Po-Wen Chung*, Yu-Chuan Lin*ACS Sustainable Chemistry & Engineering 2018, 6, 11949This study reports that the C2–C2 aldolization in ethanol conversion to C4 products, particularly butadiene, can be catalyzed by silica-supported LaMnO3 catalysts. The concentration and strength of Mn4+ was discovered to be related to the particle size of supported LaMnO3: the smaller the particle size is, the higher the concentration and acidity of Mn4+ are. The presence of high concentration and acidity of Mn4+ of small LaMnO3 particles concurrently increases the amount of weak basic nonstoichiometric oxygen, with which the surface concentration of Lewis acid–base adducts can be elevated. The Mn4+/nonstoichiometric oxygen pair is intrinsically active in C2–C2 aldolization, and the concentration of the paired site is positively correlated to the selectivity of C4 products. By coreacting ethanol with its evolved intermediates, that is, acetaldehyde and crotonaldehyde, we discovered the aldol condensation of acetaldehyde molecules to be rate-limiting. Accordingly, a plausible mechanism of aldolization of acetaldehyde molecules into C4 products mediated by the Mn4+/nonstoichiometric oxygen adduct of LaMnO3 was established.
-
Chemical transformation for 5-hydroxymethylfurfural (HMF) production from saccharides using molten salt system
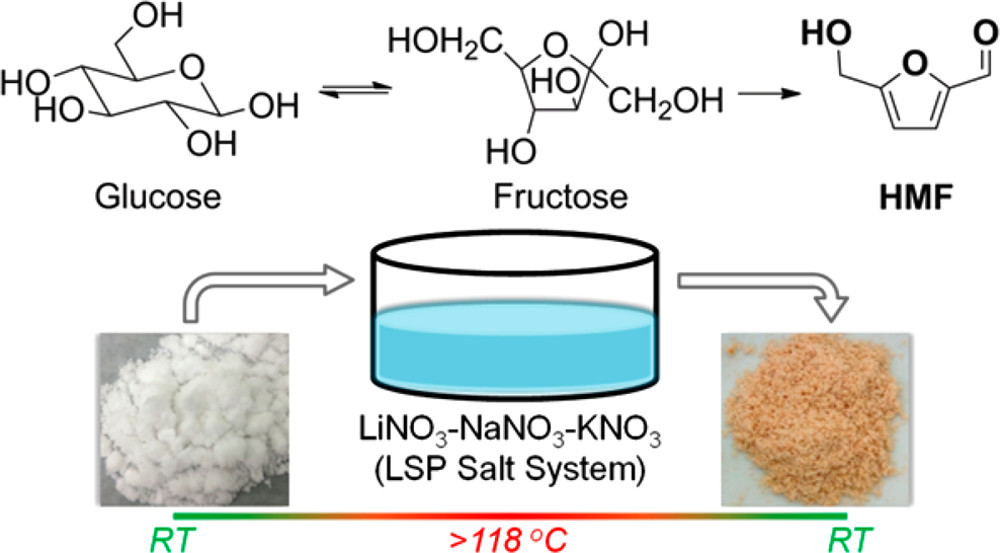
Prasenjit Bhaumik, Hao-Ju Chou, Ling-Chieh Lee, Po-Wen Chung*ACS Sustainable Chemistry & Engineering 2018, 6, 5712Herein, we have demonstrated using a eutectic ternary LiNO3–NaNO3–KNO3 (LSP) molten salt melt under mild conditions for chemical transformation of 5-hydroxymethylfurfural (HMF) production directly from biomass-derived saccharides, such as fructose, glucose, cellobiose, starch, and cellulose. In addition, 2-sec-butylphenol (SBP) was used for enhancing HMF production through efficient extraction from LSP salt melts. Notably, LSP salt can be recovered and reused without compromising activity, which could make this chemical transformation sustainable. Moreover, a simple vacuum distillation system can be employed for easy separation of HMF while processing. The aforementioned chemical transformation through LSP molten salt melts could be attributed to the intrinsic unique acid–base pair and saccharide molecule perturbation by small cations present in molten salt melts.
-
Carbon-doped SnS2 nanostructure as a high-efficiency solar fuel catalyst under visible light
Indrajit Shown, Satyanarayana Samireddi, Yu-Chung Chang, Raghunath Putikam, Po-Han Chang, Amr Sabbah, Fang-Yu Fu, Wei-Fu Chen, Chih-I Wu, Tsyr-Yan Yu, Po-Wen Chung, M.C. Lin, Li-Chyong Chen*, Kuei-Hsien Chen*Nature Communications 2018, 9, 169Photocatalytic formation of hydrocarbons using solar energy via artificial photosynthesis is a highly desirable renewable-energy source for replacing conventional fossil fuels. Using an L-cysteine-based hydrothermal process, here we synthesize a carbon-doped SnS2 (SnS2-C) metal dichalcogenide nanostructure, which exhibits a highly active and selective photocatalytic conversion of CO2 to hydrocarbons under visible-light. The interstitial carbon doping induced microstrain in the SnS2 lattice, resulting in different photophysical properties as compared with undoped SnS2. This SnS2-C photocatalyst significantly enhances the CO2 reduction activity under visible light, attaining a photochemical quantum efficiency of above 0.7%. The SnS2-C photocatalyst represents an important contribution towards high quantum efficiency artificial photosynthesis based on gas phase photocatalytic CO2 reduction under visible light, where the in situ carbon-doped SnS2 nanostructure improves the stability and the light harvesting and charge separation efficiency, and significantly enhances the photocatalytic activity.
2015
-
Importance of internal porosity for glucan adsorption in mesoporous carbon materials
Po-Wen Chung, Alexandre Charmot, Timothy Click, Yuchun Lin, YounJue Bae, Jhih-Wei Chu*, Alexander Katz*Langmuir 2015, 31,7288To better understand the adsorption of long-chain poly(1 → 4)-β-d-glucans on carbon surfaces as well as interactions responsible for this adsorption, we use a comparative study involving mesoporous carbon–silica composite materials that have been etched to varying degrees and all-atom molecular dynamics simulations. The materials synthesized as part of this etching study consist of an as-synthesized composite material (MCN-MSN), MCN-MSN-0.5 (composite materials consisting of 50% carbon by mass), MCN-MSN-0.3 (composite materials consisting of 70% carbon by mass), and MCN, in which silica etching was conducted using an aqueous ethanolic solution of either NaOH or HF. Data for the adsorption of long-chain glucans to these materials from concentrated aqueous HCl (37 wt %) solution demonstrate a direct relationship between the amount of β-glu adsorption and the magnitude of exposed carbon mesopore surface area, which systematically increases and is also accompanied by an increase in the mesopore size during silica etching. This demonstrates β-glu adsorption as occurring on internal carbon mesopores rather than exclusively on the external carbon surface. These experimental data on adsorption were corroborated by molecular dynamics (MD) simulations of β-glu adsorption to a graphene bilayer separated by a distance of 3.2 nm, chosen to correspond to the carbon mesopore diameter of the experimental system. Simulation results using a variety of β-glu solvent systems demonstrate the rapid adsorption of a β-glu strand on the graphitic carbon surface via axial coupling and are consistent with experimentally observed trends in fast adsorption kinetics. Solvent-mediated effects such as small-scale hydrophobicity and preferential interactions with ions are shown to play important roles in modulating glucan adsorption to carbon surfaces, whereas experimental data on hydrophobically modified silica demonstrate that hydrophobicity in and of itself is insufficient to cause β-glu adsorption from concentrated aqueous HCl solution.

-
Long-chain glucan adsorption and depolymerization in zeolite-templated carbon
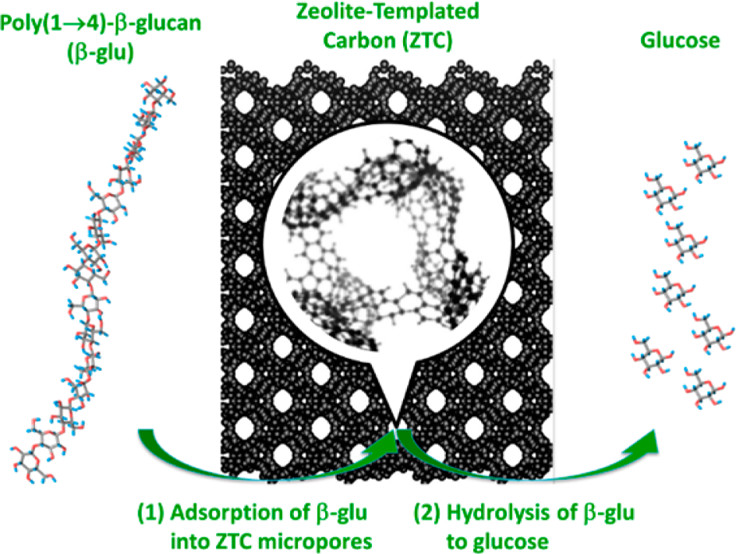
Po-Wen Chung, Mizuho Yabushita, Anh The To, YounJue Bae, Joseph Jankolovits, Hirokazu Kobayashi*, Atsushi Fukuoka*, Alexander Katz*ACS Catalysis 2015, 5, 6422We demonstrate adsorption and depolymerization of long-chain β-glu strands derived from cellulose, within the microporous confines of a zeolite-templated carbon (ZTC) material. The ZTC adsorbs β-glu strands that have a radius of gyration several-fold larger than the ZTC micropore diameter and do so rapidly (less than 2 min) and in adsorbed β-glu coverages of up to 80% of the ZTC mass. Principles of supramolecular chemistry predict that such adsorption occurs inside of the ZTC based on its micropore size as host being nearly ideal for glucan guest. A comparative study of partially etched materials and nitrogen physisorption at −196 °C indeed demonstrates β-glu adsorption to occur within internal ZTC micropores rather than on the external surface. Such adsorption under micropore confinement is expected to place significant mechanical strain on the β-glu strand, and this strain can be in principle relieved by depolymerization via hydrolysis. This hypothesis motivated us to investigate depolymerization of adsorbed β-glu strands in ZTC, where the ZTC serves as a catalyst for adsorbed β-glu hydrolysis. After a 3 h treatment in water at 180 °C, adsorbed β-glu was converted to soluble glucose in 73% yield. This represents the highest glucose yield observed to date for a carbon catalyst without postsynthetic surface functionalization and speaks to the effectiveness of weak-acid sites for β-glu hydrolysis within a constrained micropore environment.
-
Weak-acid sites catalyze crystalline-cellulose hydrolysis to glucose in water: importance of carbon post-synthetic surface functionalization
Anh The To, Po-Wen Chung, Alexander Katz*Angewandt Chemie International Edition 2015, 54, 11050The direct hydrolysis of crystalline cellulose to glucose in water without prior pretreatment enables the transformation of biomass into fuels and chemicals. To understand which features of a solid catalyst are most important for this transformation, the nanoporous carbon material MSC-30 was post-synthetically functionalized by oxidation. The most active catalyst depolymerized crystalline cellulose without prior pretreatment in water, providing glucose in an unprecedented 70 % yield. In comparison, virtually no reaction was observed with MSC-30, even when the reaction was conducted in aqueous solution at pH 2. As no direct correlations between the activity of this solid–solid reaction and internal-site characteristics, such as the β-glu adsorption capacity and the rate of catalytic hydrolysis of adsorbed β-glu strands, were observed, contacts of the external surface with the cellulose crystal are thought to be key for the overall efficiency.

Somewhat acidic: The post-synthetic functionalization of a nanoporous carbon catalyst by oxidation leads to a high density of functional weak-acid sites on the surface. These sites catalyze the hydrolysis of crystalline cellulose to glucose in high yield (70 %) and selectivity (96 %) without the need for any pretreatment.
2014
-
Catalytic hydrolysis of cellulose to glucose using weak-acid surface sites on postsynthetically modified carbon
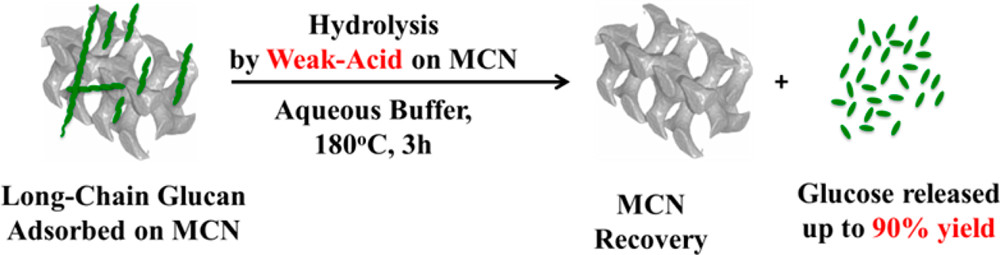
Alexandre Charmot, Po-Wen Chung, Alexander Katz*ACS Sustainable Chemistry & Engineering 2014, 2, 2866We demonstrate depolymerization of adsorbed (1 → 4)- β-d-glucans (β-glu) derived from crystalline cellulose (Avicel), using weak-acid sites of postsynthetically surface-functionalized mesoporous carbon nanoparticle (MCN) catalysts HT5-HSO3-MCN and COOH-MCN and investigate the role of acid-site density and β-glu molecular weight on this depolymerization. Both HT5-HSO3-MCN and COOH-MCN hydrolyze adsorbed β-glu strands and afford glucose yields of 73% and 90%, respectively, at a buffered pH of 2.0 after 3 h treatment at 180 °C. These yields are significantly higher than the 16% yield of an unfunctionalized MCN-control catalyst under otherwise identical conditions, demonstrating the importance of postsynthetic surface functionalization for achieving weak-acid catalytic hydrolysis. Highlighting the important role of confinement in this catalysis, all yields are generally depressed when using a lower rather than higher molecular weight of adsorbed β-glu strands on the same catalyst. The catalytic hydrolysis rate also generally increases upon decreasing buffer pH—particularly so for the more acidic carboxylic acid-functionalized catalyst COOH-MCN. This is interpreted on the basis of a higher local density of surface weak-acid sites upon protonation of surface conjugate-base functionality, as demonstrated by a comparison of zeta potential measurements of catalysts COOH-MCN and MCN.
-
Hydrolysis catalysis of miscanthus xylan to xylose using weak-acid surface sites
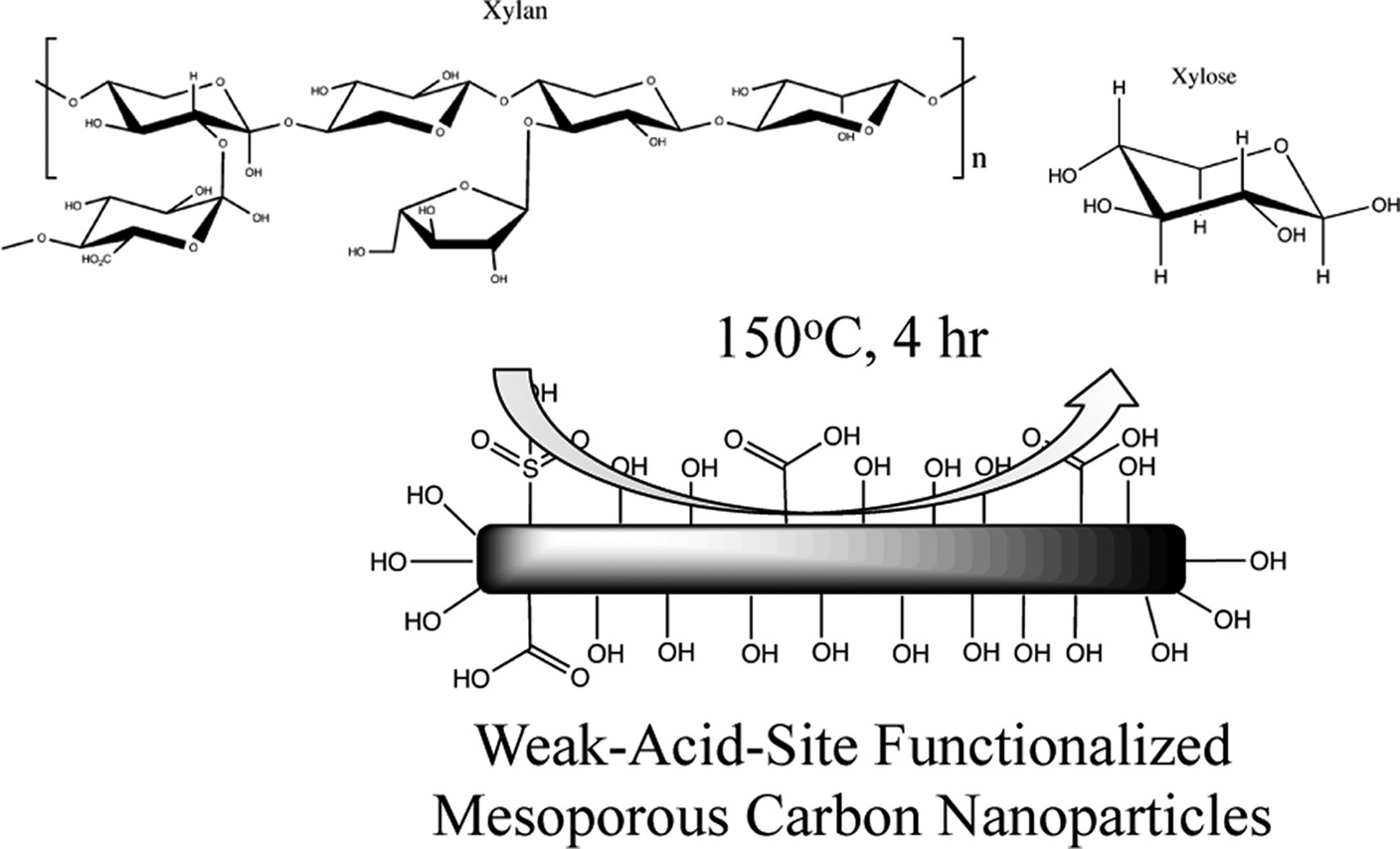
Po-Wen Chung*, Alexandre Charmot, Olayinka A. Olatunji-Ojo, Kathleen A. Durkin, Alexander Katz*ACS Catalysis 2014, 4, 302Adsorption and hydrolysis of xylan polysaccharides extracted from Miscanthus biomass are demonstrated, using surface-functionalized MCN (mesoporous carbon nanoparticle) materials that comprise weak-acid sites, at a pH corresponding to biomass extract. Extracted xylan polysaccharides consist of a peak molecular weight of 2008 g/mol according to GPC (gel-permeation chromatography), corresponding to approximately 15 xylose repeat units, and, a calculated length of 7 nm and radius of gyration of 2.0 nm based on molecular dynamics simulations. A highly active material for the adsorption and depolymerization of xylan is a hydrothermally treated sulfonated MCN material, which consists of 90% weak-acid sites. In spite of the large polysaccharide size relative to its 1.6 nm pore radius, this material adsorbs up to 76% of xylan strands from extract solution, at a weight loading of 29% relative to MCN. Starting with a 9.7% xylose yield in Miscanthus extract, this material hydrolyzes extracted xylan to xylose, and achieves a 74.1% xylose yield, compared with 24.1% yield for the background reaction in acetate buffer, at 150 °C for 4 h. Catalytic comparisons with other MCN-based materials highlight the role of confinement and weak-acid surface sites, and provide some correlation between activity and phenolic OH acid-site density. However, the lack of a directly proportional correlation between weak-acid site density and catalyzed hydrolysis rate signifies that only a fraction of weak-acid surface sites are catalytically active, and this is likely to be the sites that are present in a high local concentration on the surface, which would be consistent with previously observed trends in the hydrolysis catalysis of chemisorbed glucans on inorganic-oxide surfaces.
2012
-
Glucan adsorption on mesoporous carbon nanoparticles: effect of chain length and internal surface
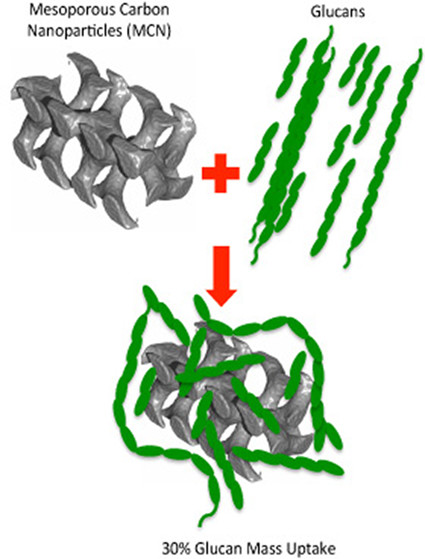
Po-Wen Chung*, Alexandre Charmot, Oz M. Gazit, Alexander Katz*Langmuir 2012, 28, 15222The adsorption of cellulose-derived long-chain (longer than ten glucose repeat units on size) glucans onto carbon-based acid catalysts for hydrolysis has long been hypothesized; however, to date, there is no information on whether such adsorption can occur and how glucan chain length influences adsorption. Herein, in this manuscript, we first describe how glucan chain length influences adsorption energetics, and use this to understand the adsorption of long-chain glucans onto mesoporous carbon nanoparticles (MCN) from a concentrated acid solution, and the effect of mesoporosity on this process. Our results conclusively demonstrate that mesoporous carbon nanoparticle (MCN) materials adsorb long-chain glucans from concentrated acid hydrolyzate in amounts of up to 30% by mass (303 mg/g of MCN), in a manner that causes preferential adsorption of longer-chain glucans of up to 40 glucose repeat units and, quite unexpectedly, fast adsorption equilibration times of less than 4 min. In contrast, graphite-type carbon nanopowders (CNP) that lack internal mesoporosity adsorb glucans in amounts less than 1% by mass (7.7 mg/g of CNP), under similar conditions. This inefficiency of glucan adsorption on CNP might be attributed to the lack of internal mesoporosity, since the CNP actually possesses greater external surface area relative to MCN. A systematic study of adsorption of glucans in the series glucose to cellotetraose on MCN shows a monotonically decreasing free energy of adsorption upon increasing the glucan chain length. The free energy of adsorption decreases by at least 0.4 kcal/mol with each additional glucose unit in this series, and these energetics are consistent with CH−π interactions providing a significant energetic contribution for adsorption, similar to previous observationsin glycoproteins. HPLC of hydrolyzed fragments in solution, 13C Bloch decay NMR spectroscopy, and GPC provide material balance closure of adsorbed glucan coverages on MCN materials. The latter and MALDI-TOF-MS provide direct evidence for adsorption of long-chain glucans on the MCN surface, which have a radius of gyration larger than the pore radius of the MCN material.
Patents
專利
USA
-
METAL OXIDES-SILICA COMPOSITE AND METHOD FOR PREPARING THE SAME
Po-Wen Chung, Meng-Xun WuU.S. Patent: US 11,511,271 B2 (2022) -
MOLTEN SALT SYSTEM AND METHOD AND APPARATUS OF TRANSFORMATION FOR MULTI-CARBON PRODUCTION BY USING THE SAME
Po-wen Chung, Prasenjit Bhaumik, Hao-Ju ChouU.S. Patent: US 10,941,130 B2 (2021) -
MODIFIED CARBONACEOUS MATERIAL, CARBON DIOXIDE ADSORBENT AND METHOD FOR CELLULOSE HYDROLYSIS USING THE SAME
Jia-Hui Wang, Hao-Ju Chou, Po-Wen ChungU.S. Patent: US 2020/0398248 A1 (2020) -
CARBONACEOUS MATERIAL FOR PURIFYING LIGNOCELLULOSIC OLIGOMERS
Po-Wen Chung, Alexandre Charmot, Alexander Katz, Amit A. GokhalU.S. Patent: US 2014/0256931 A1 (2014) -
THIOCARBONYLTHIO COMPOUND AND FREE RADICAL POLYMERIZATION EMPOLYING THE SAME
Kuo Chen Shih, Po Wen Chung, Mei Hua WangU.S. Patent: US 7,968,743 B2 (2011)
Taiwan
-
穩定化非環狀醣類複合物與穩定非環狀醣類之方法及其應用
STABILIZED ACYCLIC SACCHARIDE COMPOSITE AND METHOD FOR STABILIZING ACYCLIC SACCHARIDES AND APPLICATIONS THEREOF鍾博文, 吳佳蕙, 蔡依烜, 謝淳安中華民國專利發明號: I784687 (2022) -
熔鹽系統之用途及使用彼製造多碳產品之方法及裝置
USE OF MOLTEN SALT SYSTEM AND METHOD AND APPARATUS OF TRANSFORMATION FOR MULTI-CARBON PRODUCTION BY USING THE SAME鍾博文、布浩米克 普瑞森吉特 (Prasenjit Bhaumik)、周浩儒中華民國專利發明號: I714035 (2020) -
金屬複合氧化矽材料及其製備方法
METAL OXIDES SILICA COMPOSITE AND METHOD OF MANUFACTURING THE SAME鍾博文、吳孟勳中華民國專利發明號: I706914 (2020) -
改質碳材及使用其之二氧化碳吸附劑及纖維素水解方法
MODIFIED CARBONACEOUS MATERIAL, CARBON DIOXIDE ADSORBENT AND METHOD FOR CELLULOSE HYDROLYSIS USING THE SAME王家惠、周浩儒、鍾博文中華民國專利發明號: I704955 (2020) -
硫羰硫基化合物、以及使用此化合物之自由基聚合反應
THIOCARBONYLTHIO COMPOUND AND FREE RADICAL POLYMERIZATION USING THE SAME時國誠, 鍾博文, 王美華中華民國專利發明號: I238166 (2005)
Awards
得獎
-
Dr. Gemechu Dadi Bedasso received the Excellent Oral Presentation Award for his presentation titled “The Rational Design of Heterogeneous Catalysts for Biomass Valorization” on May 3, 2025, at the 2025 Graduate Students Symposium organized by the ACS International Chemical Sciences Chapter of Taiwan.


-
Mr. Shaw Shuo-Ren received the Excellent Oral Presentation Award for his presentation titled “Understanding the Influence of Functional Groups on CO₂ Adsorption in Carbon Materials” on May 3, 2025, at the 2025 Graduate Students Symposium organized by the ACS International Chemical Sciences Chapter of Taiwan.

-
Dr. Gemechu Dadi Bedasso awarded for Chao-Ting Chang Inorganic Chemistry Thesis Award - Honorable Mention at the 2025 Chemistry National Meeting. Thesis Title: Tailoring Carbonaceous Materials for Valorizing Biomass-Derived Feedstocks: A Substainable Route to Fine Chemicals on March 9, 2025.

-
Dr. Raju Kumar received the prestigious Academia Sinica Postdoctoral Research Fellow award for 2024 (2nd Session). His proposed project entitled "Rational Designing the Atomically Dispersed Hydrotalcite-oxide Supported Metal Catalysts for Upgrading the Bio-based Chemicals and CO2" has been recognized by Academia Sinica Postdoctoral Research Scholars, Taiwan.
-
Raju Kumar and advisor Po-Wen Chung for the 2023 Everlight Chemical Thesis Award in Green Chemistry for the Thesis Title: “Tailoring and Exploring the Surface Chemistry of Hydrotalcite-derived Oxides: Applications in Heterogeneous Catalysis” from the Chemical Society Located in Taipei (CSLT) at 2024 Chemistry National Meeting on March 29 to 31, 2024, Tamkang University, Taiwan.

-
Chih-Hsuan Tsai for awarded Chao-Ting Chang Inorganic Chemistry Thesis Award for the research achievement: Understanding the Saccharide Adsorption Dynamics Using Hydrotalcite-derived Oxides at the 2024 Chemistry National Meeting on March 31, 2024.

-
Gemechu Dadi Bedasso for IoC Excellence Award: Waste-to-wealth: Multifunctional carbon-based solid acids for cellulosic biomass valorization at Chemistry Day Poster and Oral Presentation Context on February 23, 2024 in Institute of Chemistry, Academia Sinica.

-
Raju Kumar for 2023 Significant Research Achievement: Facile Synthesis of the Atomically Dispersed Hydrotalcite Oxide Supported Copper Catalysts for the Selective Hydrogenation of 5-Hydroxymethylfurfural into 2,5-Bis(hydroxymethyl)furan on February 23, 2024 in Institute of Chemistry, Academia Sinica

-
Hsin-Hui Lee for 2023 Significant Research Achievement: Facile Synthesis of the Atomically Dispersed Hydrotalcite Oxide Supported Copper Catalysts for the Selective Hydrogenation of 5-Hydroxymethylfurfural into 2,5-Bis(hydroxymethyl)furan on February 23, 2024 in Institute of Chemistry, Academia Sinica

-
Raju Kumar et al. and advisor Po-Wen Chung for the Outstanding Research Award for research titled "A Facile Synthesis Approach: Atomically Dispersed Cuδ+ on Hydrotalcite-based Catalyst for Selective Hydrogenation of 5-Hydroxymethylfurfural to Form 2,5-Bis(hydroxymethyl)furan" at the 2023 Osaka-Kansai International Symposium in Catalysis (OKCAT2023). The symposium took place at Kyoto University in Kyoto, Japan, on November 23-24, 2023.

-
Hao-Ju Chou, Jia-Hui Wang, and advisor Po-Wen Chung for awarded TOCAT8 Poster Prize for their research achievement: Understanding the Surface Modification of Heterogeneous Catalyst for Hydrolytic Depolymerization of Cellulose: Correlation among Average Molecular Weight, Crystallinity of Polysaccharides and Hydrolytic Efficiency at 8th Tokyo Conference on Advanced Catalytic Science and Technology on August 5 to 10, 2018 in Yokohama, Japan.
-
Yang, Guo-Chuan and advisor Po-Wen Chung for awarded second place at English speech contest for their research achievement: Understanding the Depolymerization of Biomass-derived Polysaccharides: Recrystallization While Hydrolysing Polysaccharides on 36th Taiwan Symposium on Catalysis and Reaction Engineering conference hold by Catalysis Society of Taiwan on June 28 and June 29, 2018.
-
Tzeng, Tai-Wei for obtaining the subsidy from Ministry of Science and Technology for the 8th Tokyo Conference on Advanced Catalytic Science and Technology which will be held from August 5 to 10 2018 in Yokohama, Japan.